��Ŀ����
 Ϊ�ж�ij���ʴ��ڻ�ij����ǡ����ȫ��Ӧ��ͨ�����ض����ʵ���ɫ�ﵽĿ������ɫ�����ʾͳơ�ָʾ�������磺����ɫ��ʯ����ֽ����жϴ���Һ�����ԣ��õ�����Һ�����жϵ��ʵ⣨I2���Ĵ��ڣ�
Ϊ�ж�ij���ʴ��ڻ�ij����ǡ����ȫ��Ӧ��ͨ�����ض����ʵ���ɫ�ﵽĿ������ɫ�����ʾͳơ�ָʾ�������磺����ɫ��ʯ����ֽ����жϴ���Һ�����ԣ��õ�����Һ�����жϵ��ʵ⣨I2���Ĵ��ڣ���֪����SO2+I2+2H2O�TH2SO4+2HI����ɫ��Һ����
�ﳣ���£�������ˮ��
��5SO2+2H2O+KMnO4�TK2SO4+2H2SO4+2MnSO4����ɫ��Һ����
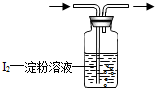
��1����ҵ�ϲ���SO2��N2��O2���������SO2��������ͼ����װ�ã�����װ��ʡ�ԣ���
�ٻ������ͨ��ͼʾ����װ��һ��ʱ�����Һ��ɫ����ɫ��Ϊ
��
��
ɫʱ����ֹͣͨ�����˷����˲�SO2��������
��
��ѡ��͡��ߡ����Ļ�����壮������װ���ڵ�I2�ĵ�����ҺҲ������
�������
�������
��Һ������Ϊָʾ������Ϊ���������Һ����һ������SO2�����Ϻ�ɫ��Ϊ��ɫ
���������Һ����һ������SO2�����Ϻ�ɫ��Ϊ��ɫ
����������������Һ�ı�ɫ����жϷ�Ӧ�Ľ���״̬�����Է�Ӧ���е�ʱ�����п��ƣ����ݴ�װ�������յĶ�������������������������ж����������ߵ͵����⣻
����������й��ڸ��������Һ���������ķ�Ӧ���з������ɣ���
����������й��ڸ��������Һ���������ķ�Ӧ���з������ɣ���
����⣺�ٵ�ĵ�����Һ����ɫ������SO2+I2+2H2O�TH2SO4+2HI����ɫ��Һ����֪����Һ����ɫ�����ɫʱ�÷�Ӧǡ����ɣ���ʱӦ��ֹͣͨ�����壬ͨ����������ʹ���������ȷ�������£�������ˮ�����Ե�ĵ�����Һ���ն������������С���ʸ�װ�����ն������������С�����Դ˷����˲�SO2�����ϵ͵Ļ�����壬����ޣ��ͣ�
����Ϊ�Ϻ�ɫ������ص���Һͨ���������ʱ�ᷢ��5SO2+2H2O+KMnO4�TK2SO4+2H2SO4+2MnSO4����ɫ��Һ���ķ�Ӧ����Һ�������Ϻ�ɫ�����ɫ������Ҳ�ɲ��ø��������Һ�����ĵ�����Һ��ɸ�ʵ�飬���������أ����������Һ����һ������SO2�����Ϻ�ɫ��Ϊ��ɫ��
����Ϊ�Ϻ�ɫ������ص���Һͨ���������ʱ�ᷢ��5SO2+2H2O+KMnO4�TK2SO4+2H2SO4+2MnSO4����ɫ��Һ���ķ�Ӧ����Һ�������Ϻ�ɫ�����ɫ������Ҳ�ɲ��ø��������Һ�����ĵ�����Һ��ɸ�ʵ�飬���������أ����������Һ����һ������SO2�����Ϻ�ɫ��Ϊ��ɫ��
������������һ��ʵ��̽���ۺ��⣬����Ĺؼ��dz������������ṩ������Ϣ��ɣ������漰������ϸߵ����ϵͣ�

��ϰ��ϵ�д�
�����Ŀ
 ��2012?���ݣ�Ϊ�ж�ij���ʴ��ڻ�ij����ǡ����ȫ��Ӧ��ͨ�����ض����ʵ���ɫ�ﵽĿ������ɫ�����ʾͳơ�ָʾ�������磺����ɫ��ʯ����ֽ����жϴ���Һ�����ԣ��õ�����Һ�����жϵ��ʵ⣨I2���Ĵ��ڣ�
��2012?���ݣ�Ϊ�ж�ij���ʴ��ڻ�ij����ǡ����ȫ��Ӧ��ͨ�����ض����ʵ���ɫ�ﵽĿ������ɫ�����ʾͳơ�ָʾ�������磺����ɫ��ʯ����ֽ����жϴ���Һ�����ԣ��õ�����Һ�����жϵ��ʵ⣨I2���Ĵ��ڣ�

