��Ŀ����
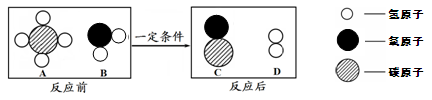
ʵ��С���о����ᡢ�������ơ����������������ʵĻ�ѧ���ʣ�������ͼ��ʾ��ʵ�顣��������ͼ������ҩƷ������һ������о���
��1��ʵ���ij�Թ���Ϊ��ɫ��Һ�����Թ��з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2��ʵ���ij�Թ���Ϊ��ɫ��Һ�������м��������� ����Һ��Ϊ��ɫ���ɴ��ƶϣ����Թ������ʢ�е������� ��
��3��ʵ���ij�Թܵײ�����ɫ��״���������������м��������� ���������ʧ��
��4��ʵ���ij�Թܵĵײ��а�ɫ���壬���˺�����Һ�еμ�ϡ���ᣬ��ʼʱ����������һ��ʱ��������ݳ��֡��ɴ��ƶϣ����Թ������������Ӧ�Ļ�ѧ����Ϊ ��
��5��ʵ���ij�Թ���ֻ�õ���ɫ��Һ�������м���������̼������Һ��ֻ�������ݡ���ô���Թ������������Ӧ�Ļ�ѧ����Ϊ ��
��6��ʵ�����ʱ�����ǽ�ʵ���Һ����ͬһ��Һ���У��۲�����Dz������յķ�Һ���ܳ����ԡ�Ϊ����֤���룬��������ʵ����δ�漰������Ļ�ѧ���ʣ���ѡҩƷ����ʵ�飺ȡ������Һ��Ʒ���Թ��У������м���һ������ ���۲�����֤�����յķ�Һ�����ԡ�ʵ���Ϊ�˱����Һ��ɲ������ �����ǶԷ�Һ������ǡ��������
��1��Fe2O3+6HCl=2FeCl3+3H2O ����2��ϡ���� �� ��ɫ��̪��Һ �� ��3��ϡ���
��4��Ca(OH)2+Na2CO3=CaCO3��+2NaOH����5��Na2CO3+2HCl=2NaCl+H2O+ CO2������6�����ۣ��𰸺������ɣ�
���������������1����������ϡ���ᷴӦ�����Ȼ�����ˮ���Ȼ�������ˮ�ʻ�ɫ��д����Ӧ����ʽ����2�������⡰��ɫ��Ϊ��ɫ���DZ�Ϊ��ɫ���ɵã�һ��������ɫ��̪��Һ�м����˼ʵ�������������£���3������ɫ��״������Ϊ������ͭ��������������У��ʼ��������м���������ϡ���ᣬ��ʹ������ʧ����4�����ײ��а�ɫ���壬��ʼʱ����������һ��ʱ��������ݳ��֡���˵����Һ����̼������ڣ���ʼʱ��������Һ�е���һ�����ʷ�Ӧ�������Ϊ����������̼���Ƶķ�Ӧ����5��������������̼������Һ��ֻ�������ݡ���˵��������ڣ���Ϊ������̼���Ƶķ�Ӧ����6������ʵ�����漰���������У����ָʾ�����ᡢ��ķ�Ӧ������������������Σ�û���漰��������ķ�Ӧ��ѡ�ó����Ļ��ý������ɡ�
���㣺̽��������Ҫ����

���꣬���ݡ��Ӵ��ס��¼��ٴ����������Ƕ�ʳƷ��ȫ��������Ⱦ�����˼����С��ͬѧ�������ӵ������Ϣ������Ԫ��λ�ڵ������ڢ���塣����������ɫ�й���Ľ������۵�320.9�棬�е�765�棬�ܶ�8.64g/cm3�������Ժ���չ�ԣ����ڳ�ʪ�����л���������ʧȥ���������ӿ������ᣬ�������ڼ���Ӷ�����ʮ���к��������������Ⱦ��ʳƷ��ˮ��������κ����༲�����ش��������⣺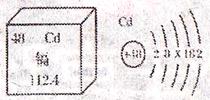
��1������ͼ�е������Ϣ�����ж���Ԫ�ص��жϲ���ȷ����
| A�������ڽ���Ԫ�� | B��һ����ԭ������48������ |
| C����ԭ��ʾ��ͼ��X=18 | D���ӵ����ԭ������Ϊ112.4g |
��3�����Ӵ��ס���Ҫ�����ں�ˮ�����������Ⱦ�ĵ������������Ӵ��ס���Ҫ��Դͷ������Ŀǰ��ˮ�����ķ����ǣ�������Na2CO3Ͷ�뱻Cd2+��Ⱦ�ĺ�ˮ�У��γ�CdCO3�������Խ��ͺ�ˮ��Cd2+��Ũ�ȡ�
����ij�Ӷ���Ҫ�����Ȼ��ӣ���̼���Ʒ�Ӧ�Ļ�ѧ����ʽΪ ��
��25��ʱ��CdCO3���ܽ��Ϊ2.8��10-6g����1L��ˮ�к�Cd2+������Ϊ mg����ˮ�ܶ�Ϊ1.0g/cm3����������ȷ��0.001mg��������Ҫ��ˮ��Cd2+�ĺ����ı��Q0.005mg/L����Na2CO3�����ĺ�ˮ ����ǡ�����ꡣ
��������ͭ�������ֽ�����ij�о�С���ͬѧΪ̽������ͭ�����Ľ������˳�����������ʵ�飺��.����Ƭ����ϡ�����У���.����Ƭ����ϡ�����У���.����Ƭ��������ͭ��Һ�С�
(1)ʵ���������ǣ������ݲ�������Һ����ɫ��Ϊdz��ɫ����Ӧ�Ļ�ѧ����ʽΪ��_____________________________________________________��
��Ӧ�������������________�ķ������顣
(2)��������ʵ�黹������ȫ֤�����ֽ����Ļ��˳�����㲹��һ��ʵ�����ﵽʵ��Ŀ��(д��ʵ�����������)____________________________��
���������о������ֽ����Ļ����ǿ������˳����____________��
(3)������ͭ�����е����ֽ����ֱ����________��Һ�У�������֤�����ֽ����Ļ��ǿ����
(4)��ͭƬ���ȣ�ͭƬ�����ڡ�ͬѧ���������ʵ��֤���˺�ɫ����������ͭ��
| ��ʵ����� | ��ʵ������ |
| ����һƬ��ڵ�ͭƬ�������Թ��У�����������______��Һ | ͭƬ����ĺ�ɫ����ȫ����ʧ��¶����ɫ��ͭ����Һ��Ϊ________ɫ |
��9�֣���2013?����һģ��С���Ķ��������ϵ�֪������صķֽ���ö������̡�����ͭ�����������������ǣ�����Ӱ������طֽ�����ؼ������Ĵ�Ч��������̽����Ȥ��
��������⡿����ͭ�Ƿ�ȶ������̴�Ч�����ã�Ӱ������طֽ����ʵ���������Щ�أ�
�����ʵ�顿С�������ɵ����������Ϊ������������м���ʵ�飮
| ��� | KClO3������ | �������ʵ����� | �¶� | ��������� | ��Ӧ����ʱ�� |
| �� | 2.0g | | 330�� | 100mL | t1 |
| �� | 2.0g | CuO 0.5g | 330�� | 100mL | t2 |
| �� | 2.0g | MnO2 0.5g | 330�� | 100mL | t3 |
| �� | 2.0g | MnO2��_________�� g | 380�� | 100mL | t4 |
��2������ΪС�����ʵ��ۺ�ʵ��ڶԱȵ�Ŀ������_________����
��3��ʵ�����MnO2������Ϊ��_________��g����t3��t4����ѧ��Ӧ�������¶ȵĹ�ϵ����_________����
��4��д��ʵ������漰�Ļ�ѧ����ʽ��_________����
��������ʵ���Ͳ���ȷ����
| ѡ�� | �� ʵ | �� �� |
| A | һ��ˮ�д�Լ��1��67��1021��ˮ���� | ���Ӻ�С |
| B | ���������еľƾ����� | �����Dz����˶��� |
| C | ���¼��е�ˮ���������������� | ԭ�ӵ������������ |
| D | ���ȵ��������г���̥���ױ��� | �����¶ȸߣ����Ӽ����� |
�κλ�ѧ��Ӧ��Ҫ���������غ㶨�ɣ�����˵�����������غ㶨�ɵ��ǣ�������
| A����˿��������ȼ�����ɹ������������ԭ����˿������ |
| B��2g������8g������ȼ����ַ�Ӧ��Ȼ����10g��ˮ |
| C����ּ��ȸ�����غ���������������ԭ��������ص����� |
| D��ij������������ȼ�����ɶ�����̼��ˮ��˵����������һ������̼���⡢������Ԫ�� |