��Ŀ����
�ú��ж�����̼��ˮ�������ʵ�ij�ֻ�ԭ������ⶨһ�����������FexOy������ɣ�ʵ��װ����ͼ��ʾ��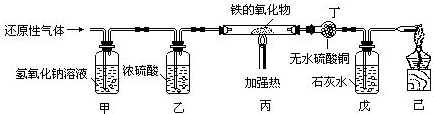
������ͼ�Իش�
��1����װ�õ�������
��2��ʵ������ж�װ��û�����Ա仯������װ������Һ�����˰�ɫ��������û�ԭ��������
��3������װ���е�FexOy��ȫ������ԭ����ʣ����������Ϊ16.8g��ͬʱ�����װ�õ�����������17.6g���������������Ļ�ѧʽΪ
��4������ʵ��װ���У����û�м�װ�ã���ʹ�ⶨ�������Ԫ������Ԫ�ص������ı�ֵ
��������Ϊ�ۺ�ʵ�������û�ԭ������ⶨ�������������ɣ���ԭ���Ǹ��������������ͨ������ȷ��������ͳ��нΣ���ԭ��������������һ����̼�����Ƕ���������ˮ�Ͷ�����̼��ԭ�����л��ж�����̼��ˮ���������Ա����ȳ�ȥ��Ȼ���û�ԭ�����巴Ӧ������װ�����Լ������ɵ�ˮ�Ͷ�����̼�����ļ�װ�ý���β���������л������
����⣺��1���������̼�����������Ʒ�Ӧ������ʽΪ��2NaOH+CO2�TNa2CO3+H2O�����Գ�������̼������������Һ����Ũ���������ˮ�ԣ����Գ�ˮ����ѡ��Ũ���ᣮ��������̼ʱ������Ҫ����Һ��ͨ���������ˮ����������Ҫ�ȳ�������̼�ٸ��˵�Լ�װ�õ������dz�ȥ�������գ�������̼��
��2����װ��ʢ�ŵ�����ˮ����ͭ����ˮ����ͭ��ˮ���������װ��û�����Ա仯��˵��û��ˮ���ɣ��ʸû�ԭ�����岻����������װ��ʢ�ŵ���ʯ��ˮ��������̼��ʹʯ��ˮ����ǣ���װ������Һ�����˰�ɫ�������ʸû�ԭ��������һ����̼��
��3����װ���е�FexOyȫ������ԭ��ʣ�����Ϊ���ɵ�������������װ����ʢ�ŵ�ʯ��ˮ��ˮ������̼�����ӵ�����Ϊ������̼��������
YCO+FeXOY
XFe+YCO2
56X 44Y
16.8g 17.6g
=
��ã�
=
���Ը���������������Ϊ��Fe3O4
��4�����û�м�װ�ã����еĶ�����̼Ҳ������װ�ñ����գ��ʶ�����̼������ƫ���൱��һ����̼��õ������������ӣ��ʲ�ý������Ԫ������Ԫ�ص������ı�ֵƫС��һ����̼�ж�������Ⱦ���������ļ�װ�ö�β�����д��������л������
�ʸ���Ĵ�Ϊ��
��1������CO2��2NaOH+CO2�TNa2CO3+H2O
��2��CO����һ����̼��
��3��Fe3O4
��4��ƫС���ж���һ����̼����Ⱦ������
��2����װ��ʢ�ŵ�����ˮ����ͭ����ˮ����ͭ��ˮ���������װ��û�����Ա仯��˵��û��ˮ���ɣ��ʸû�ԭ�����岻����������װ��ʢ�ŵ���ʯ��ˮ��������̼��ʹʯ��ˮ����ǣ���װ������Һ�����˰�ɫ�������ʸû�ԭ��������һ����̼��
��3����װ���е�FexOyȫ������ԭ��ʣ�����Ϊ���ɵ�������������װ����ʢ�ŵ�ʯ��ˮ��ˮ������̼�����ӵ�����Ϊ������̼��������
YCO+FeXOY
| ||
56X 44Y
16.8g 17.6g
| 56x |
| 44y |
| 16.8g |
| 17.6g |
| X |
| Y |
| 3 |
| 4 |
���Ը���������������Ϊ��Fe3O4
��4�����û�м�װ�ã����еĶ�����̼Ҳ������װ�ñ����գ��ʶ�����̼������ƫ���൱��һ����̼��õ������������ӣ��ʲ�ý������Ԫ������Ԫ�ص������ı�ֵƫС��һ����̼�ж�������Ⱦ���������ļ�װ�ö�β�����д��������л������
�ʸ���Ĵ�Ϊ��
��1������CO2��2NaOH+CO2�TNa2CO3+H2O
��2��CO����һ����̼��
��3��Fe3O4
��4��ƫС���ж���һ����̼����Ⱦ������
��������ʵ���Ŀ�����û�ԭ������ⶨ�������������ɣ����Ĺؼ��Ƿ���ʵ��װ�ã��˽�ÿһ�������ã�

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ
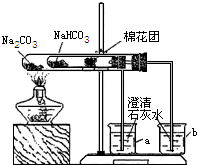 ijУͬѧ���г��ϵ�ʳƷ���ɼ�����������̽����
ijУͬѧ���г��ϵ�ʳƷ���ɼ�����������̽����ʵ��һ����С���ͬѧ�ռ���һ����Ҫ�ɷ���С�մ�̼�����ƣ������ɼ�
��1��ȡ��Ʒ����������ˮ�У������Һ��pH����7��
��2����С�մ���Ʒ�еμ����ᣬ�д������ݲ�����������������Ƕ�����̼���÷�Ӧ�Ļ�ѧ����ʽΪ
��3��ͬѧ��ͨ���������ϣ�����ʦ��ָ���²�������ͼ��ʾװ����С�մ�ʹ������˶Ա�ʵ�飮ʵ����С�Թ���մ����ˮ����ͭ��ĩ����������ɫ��a�ձ���ʯ��ˮ����������
b�ձ���ʯ��ˮ���ְ�ɫ���ǣ��Իش𣺢���ʵ�����ʱ��������Ҫע��������ǣ�
����ʵ�飨3�����Եó��Ľ�����
ʵ�������С���ͬѧ�ռ���һ����Ϊ�����ۡ������ɼ���Ϊ̽������ɣ�����ʵ�飺
��1�������ۡ�Ϊһ�ְ�ɫ���壬������ˮ��
��2��ȡ������Ʒ��ϡ�����Ϻ�����˿���ʹʯ��ˮ����ǵ����壻
��3��ȡ������Ʒ���Ⱥ������д̼�����ζ�����壻
��4��������Ʒ������������Һ��Ϻ��ȣ�������ʹʪ��ĺ�ɫʯ����ֽ�����ij�����
������֪�������ۡ�����Ҫ�ɷ��dz��л�ѧ�α���һ�ֳ������Σ�
������������
���á����ۡ������ɼ�����ըʳƷ���ú������ۺ�ʳ�õ�ԭ�������
��ijЩС�̷�ϲ��ѡ�á����ۡ����桰С�մ������ɼ���ԭ�������
ʵ��������С��ͬѧ̽��С�մ����ۡ�������ʳƷ�����в������ݵ�ԭ��
��1���������ɼ������������ж�����
��2��С���ͬѧ��������ۼ���ˮ��ȡ�ϲ�Һ����pH��ֽ��ã�pH=7���²���������ԭ����
ʵ���ģ���С���ͬѧ��ij���з���һ�ָ������ɼ�--���ͷۣ���ɷּ�����
| �ɷ� | �������� |
| ̼������ | ̼������ |
| �������� | �����ᡢ��ʯ��� |
| ���ɼ� | ���� |
| �����ɷ� | ���ۡ�֬����� |
��2��Ϊ�ⶨ����̼�����Ƶ�����������С���ͬѧ���������ʵ�飬����Ҫʵ�鲽�����£�
��ͼ��װ��������50.0g����������ƿ�У���������ij����Һ��
����һ��ʱ���ڸ�������������ӵ����������������ߣ���
����ƿ�в��ٲ�������ʱ������B���ӵ���A����������һ�����Ŀ�����
�����ٴγ�����������������ӵ�������
�����ظ���͢��IJ�����ֱ��������������������䣮
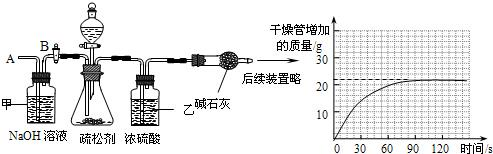
���ۣ�
�ٲ��������Ʒǰ��Ӧ���
��װ���ҵ�������
���������й����ݼ���������̼�����Ƶ�������������д��������̣���С��4�֣�
 �������һ������ۣ�����Ҫ�ɷ�̼�����ƣ�NaHCO3�����ƻ�������ά�ṹ���ٽ���������ˮ�֣��Ӷ�ʹ�����������ﵽ����ˬ����Ŀ�ģ�С��ֽ����˰����۵�����ŵ����в�����ʳ�ȵ�������ʱ�����˴������壬���Դ��ĸ���Ȥ�������������̽����
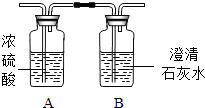
�������һ������ۣ�����Ҫ�ɷ�̼�����ƣ�NaHCO3�����ƻ�������ά�ṹ���ٽ���������ˮ�֣��Ӷ�ʹ�����������ﵽ����ˬ����Ŀ�ģ�С��ֽ����˰����۵�����ŵ����в�����ʳ�ȵ�������ʱ�����˴������壬���Դ��ĸ���Ȥ�������������̽������С�ڶ�����ijɷֽ����˲²⣺�����г��˴�������֮�⣬�����ܺ���ˮ������CO2�����跨�ռ�������Ʒ����ȥ�������������ͼʵ��װ�ý���̽��������������ͨ��A��B���Լ�ƿ����֪Ũ���������ˮ�ԣ�
ʵ������Aƿ�������ӡ�Bƿ���а�ɫ�����������
ʵ����ۣ���������һ������
��С�ڶԶ�����̼����Դ�������ֲ²⣺
�²�٣�
�²�ڣ�������̼��������̼���������ȷֽ�����ģ�
������ϲ²⣬С�����������ʵ�飺������������б����еĿհף�
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| ʵ��1 | �������ȷ | ||
| ʵ��2 | ȡ̼��������Ʒ���������Թ��У����ȣ����Ѳ���������ͨ�������ʯ��ˮ | �Թܿ���Сˮ�β�������ʹ�����ˮ��ˮ����ǵ�������� | �������ȷ |
�������ϣ��ڰٶ���ҳ������̼�����ƵĻ�ѧ���ʣ����Ի�ã�̼�����������ֽ⣬��������̼�������ɣ��ٸ���ʵ��2д��̼���������ȷֽ�Ļ�ѧ����ʽ
����ʵ��2���а�ɫ��ĩʣ�࣬����������������֤�䲻���������ƹ��壬�û�ѧ��������֤�����̼���ƹ��壻
��1������������
��2����ѧ������
��������Ӧ�ã�������ͷʱ������ͨ���ڷ��ͺ�������м���С�մ�NaHCO3��������NaHCO3��Ŀ����
 ���ǵ����������������ƣ��ִ�������������Ļ�ѧ֪ʶ��
���ǵ����������������ƣ��ִ�������������Ļ�ѧ֪ʶ��