��Ŀ����
ʵ����������1000g������������Ϊ4%������������Һ��
�����ף����ò���Ϊ��
��1�����㣺���������ƹ��� g��ˮ mL��ˮ���ܶ���1g/cm3����
��2��������������Ϊ23.1g���ձ���������������������ƽ�ϳ�ȡ�������ƹ���ʱ��ʢ���������ƹ�����ձ�Ӧ������ �̣�����ҡ��������±����е������У�ѡ���������루�̡���ʾѡ�ã���
�������б����ѡ������ȷ��ʾ����λ�õ�ѡ�� ������ĸ��

��3���ܽ⣺���������ƹ�������ˮ���� �����裬ʹ��������ȫ���ܽ⣬��ȴ�����£�
��4������õ���Һװ���Լ�ƿ���Ǻ�ƿ�Dz����ϱ�ǩ�������Լ����У�
�����ù����У���������������������С��4%�Ŀ���ԭ���� ��дһ�֣�
��5������ʦ�ṩ��ҩƷ��500g8%������������Һ��500g1%������������Һ���������������ƹ����ˮ�������������÷����⣬��������Ƴ��������÷�����д������ҩƷ�����Ƽ�������
�ң� ����
��6�����������漰�ķ�������ѷ���Ϊ ������ ��
�����ף����ò���Ϊ��
��1�����㣺���������ƹ���
��2��������������Ϊ23.1g���ձ���������������������ƽ�ϳ�ȡ�������ƹ���ʱ��ʢ���������ƹ�����ձ�Ӧ������
| ����/g | 100 | 50 | 20 | 20 | 10 | 5 |
| �̡���ʾѡ�� |

��3���ܽ⣺���������ƹ�������ˮ����
��4������õ���Һװ���Լ�ƿ���Ǻ�ƿ�Dz����ϱ�ǩ�������Լ����У�
�����ù����У���������������������С��4%�Ŀ���ԭ����
��5������ʦ�ṩ��ҩƷ��500g8%������������Һ��500g1%������������Һ���������������ƹ����ˮ�������������÷����⣬��������Ƴ��������÷�����д������ҩƷ�����Ƽ�������
�ң�
��6�����������漰�ķ�������ѷ���Ϊ
���㣺һ������������������Һ������,������-������ƽ
ר�⣺��Һ����Һ���ܽ��
��������1��������������������Һ�������������������ij˻����з�����
��2������ʹ��������ƽ��ȡ�������������IJ����������������롢�����ѡ�á�����ĵ����������
��3���ܽ�������ò��������裬���ٹ����ܽ⣻
��4��������Һ������������������С��4%�Ŀ���ԭ���ǣ��������ˣ�����������������������ʣ����벻����λ�þ͵�����ƽƽ�⣩���ܼ����ˣ���ˮʱ���Ӷ������ձ����Լ�ƿ����ˮ�����ݴ˷�����
��5������6��������Һ�ķ����ࣺܶ��ֱ�������ʺ�ˮ�䣻�������������Һ��ʱ��Ҫ����һ�������Ƿ�������������ʣ�ʣ���������ˮ���䣮
��2������ʹ��������ƽ��ȡ�������������IJ����������������롢�����ѡ�á�����ĵ����������
��3���ܽ�������ò��������裬���ٹ����ܽ⣻
��4��������Һ������������������С��4%�Ŀ���ԭ���ǣ��������ˣ�����������������������ʣ����벻����λ�þ͵�����ƽƽ�⣩���ܼ����ˣ���ˮʱ���Ӷ������ձ����Լ�ƿ����ˮ�����ݴ˷�����
��5������6��������Һ�ķ����ࣺܶ��ֱ�������ʺ�ˮ�䣻�������������Һ��ʱ��Ҫ����һ�������Ƿ�������������ʣ�ʣ���������ˮ���䣮
����⣺��1�����������ƹ�����������1000g��4%=40g��
ˮ������Ϊ��1000g-40g=960g��
��Ҫˮ�����=960g��1g/cm3=960cm3=960mL��
��2����ȡ��������ʱΪ��ֹ��ʴ��ƽ�����̣������ձ��ڸ��ݡ��������롱��ԭ�������������ڽ��г�ȡ���������ƹ������ձ���������Ϊ40g+23.1g=63.1g������ѡ������ʱӦѡ��50g��10g�����һ��������Ӧ�ƶ���3.1g�����������ʾ��Ϊ����������ͼ��A��ʾ2.9g��B��ʾ3.1g��C��ʾ3.0g��
��3���ܽ���������ڽ����Լӿ������ܽ������Ϊ��������
��4��������Һ������������������С��4%�Ŀ���ԭ�������֣������ٻ��ܼ��࣬�������ˣ�����������������������ʣ����벻����λ�þ͵�����ƽƽ�⣩���ܼ����ˣ���ˮʱ���Ӷ������ձ����Լ�ƿ����ˮ����
��5��500 g 8%������������Һ�к����ʣ�500 g��8%=40g����ˮ��1000g-500g=500g�����Կ�����500 g 8%������������Һ��500gˮ�����ƣ�
400 g 1%������������Һ�к����ʣ�400 g��1%=40g����ˮ��1000g-400g=600g�����Կ�����400 g 1%������������Һ��600gˮ�����ƣ�
��6�������ң�ֻ�����500 gˮ������������������Ҫ��������������Һ��������ˮ��������
�ʴ�Ϊ����1��40��960��
��2����
B��
��3����������
��4������ʱ�������ƺ�����ŷ��ˣ���������������ɣ���
��5��500 g 8%������������Һ��500gˮ�����ƣ�400 g 1%������������Һ��600gˮ�����ƣ�
��6���ң�������㣬��С��
ˮ������Ϊ��1000g-40g=960g��
��Ҫˮ�����=960g��1g/cm3=960cm3=960mL��
��2����ȡ��������ʱΪ��ֹ��ʴ��ƽ�����̣������ձ��ڸ��ݡ��������롱��ԭ�������������ڽ��г�ȡ���������ƹ������ձ���������Ϊ40g+23.1g=63.1g������ѡ������ʱӦѡ��50g��10g�����һ��������Ӧ�ƶ���3.1g�����������ʾ��Ϊ����������ͼ��A��ʾ2.9g��B��ʾ3.1g��C��ʾ3.0g��
��3���ܽ���������ڽ����Լӿ������ܽ������Ϊ��������
��4��������Һ������������������С��4%�Ŀ���ԭ�������֣������ٻ��ܼ��࣬�������ˣ�����������������������ʣ����벻����λ�þ͵�����ƽƽ�⣩���ܼ����ˣ���ˮʱ���Ӷ������ձ����Լ�ƿ����ˮ����
��5��500 g 8%������������Һ�к����ʣ�500 g��8%=40g����ˮ��1000g-500g=500g�����Կ�����500 g 8%������������Һ��500gˮ�����ƣ�
400 g 1%������������Һ�к����ʣ�400 g��1%=40g����ˮ��1000g-400g=600g�����Կ�����400 g 1%������������Һ��600gˮ�����ƣ�
��6�������ң�ֻ�����500 gˮ������������������Ҫ��������������Һ��������ˮ��������
�ʴ�Ϊ����1��40��960��
��2����
| ����/g | 100 | 50 | 20 | 20 | 10 | 5 |
| �̡���ʾѡ�� | �� | �� |
��3����������
��4������ʱ�������ƺ�����ŷ��ˣ���������������ɣ���
��5��500 g 8%������������Һ��500gˮ�����ƣ�400 g 1%������������Һ��600gˮ�����ƣ�
��6���ң�������㣬��С��
�����������ϸ�µĿ�������ƽ��ʹ�ú���Һ���Ƶ���������֪ʶ���ܽ�����ʵ�����������ȷ���������������ĺ���ǽ��Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
���в����ɵ�����������ȼ�ն��õ��������ǣ�������
| A��P2O5 |
| B��Fe2O3 |
| C��CO2 |
| D��SO2 |
���з�Ӧ������������Ӧ���������ڻ��Ϸ�Ӧ���ǣ�������
A���������
| ||
B����+����
| ||
C������+��
| ||
D���ƾ�+����ˮ
|
���б仯�ǻ�ѧ�仯���ǣ�������
| A������Һ�� | B��þ��ȼ�� |
| C��ˮ��� | D��ľ���Ƴ����� |

 �ס��ҡ������ֹ������ʵ��ܽ��������ͼ��ʾ����ش�
�ס��ҡ������ֹ������ʵ��ܽ��������ͼ��ʾ����ش�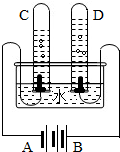 �������ĿҪ��ش��������⣮
�������ĿҪ��ش��������⣮