��Ŀ����
�������������������Ӧ��ʮ�ֹ㷺��
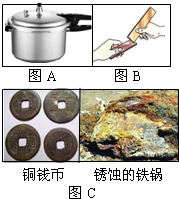
��1����ͼ�ǽ���ͭ��һ��Ӧ��ʵ���������˽���ͭ��_______�ԡ�
��2������Ʒ�ڿ����в�����ʴ��ԭ�������С����ұ��������ã��������ܵı���Ĥ�����ڿ����С����ұ�������Ӧ�Ļ�ѧ����ʽΪ______________��
��3������Ʒ��������ʴ��
��ijͬѧ�������ͼ��ʾ��װ��̽������ʴ��������a����װ��������е�����ˮ��b�����Ǹ���Ŀ���������L����R�˵�Һ��߶�һ�£�����ľ����һ��ʱ���۲졣�����ƶϴ������ ��
| A��ֻ��a���е���������ʴ |
| B��L����R�˵�Һ��߶���Ȼ����һ�� |
| C����ʵ�鲻��˵��������������й� |
| D����ʵ��˵��������ʴ�������ˮ�й� |
��4����ֻ��һ�ֽ������ʣ���������Һ������֤п��ͭ�������ֽ������˳������֤���������漰�Ļ�ѧ����ʽΪ____________________________������˿������ͭ��Һ��Ӧ��ɡ�ͭ����ʵ��ʱ����Ҫ��ȥ��˿���������Ĥ�����õ�����������__________������ϡ���ᴦ����������_________����ʱ����������Ĥ�ѱ��ƻ���
��5���ϳ�����CO��H2���������Ʊ�����Ϊ21��������ȼ�ϵĶ����ѣ�CH3OCH3������������ұ���������䲿����������ʾ��ͼ���£�

�ٶ����ѣ�CH3OCH3�����ɺϳ�����CO��H2����һ�����������Ƶá���Ӧ������CO��H2�����Ӹ�����1��2���з�Ӧ���Ƶö�����ʱ�����ɵ���һ�������� ��
�ڸ���ʾ��ͼ�б�ʾ�����ʣ��ϳ�����ұ���������������з�����������Ҫ�ķ�Ӧ�����в������û���Ӧ�Ļ�ѧ����ʽΪ ��
��1�������� ��2��4Al+3O2====2Al2O3 ��3���� B C ���Ƴɲ���֣����ƳɺϽ� ��4�� Cu+2Ag(NO3)3=====Cu(NO3)2+2Ag ��ɰֽ��ĥ
�����ݲ��� ��5����H2O ��3CO+Fe2O3 ���� 2Fe+3CO2
����

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

 �������������������Ӧ��ʮ�ֹ㷺��
�������������������Ӧ��ʮ�ֹ㷺��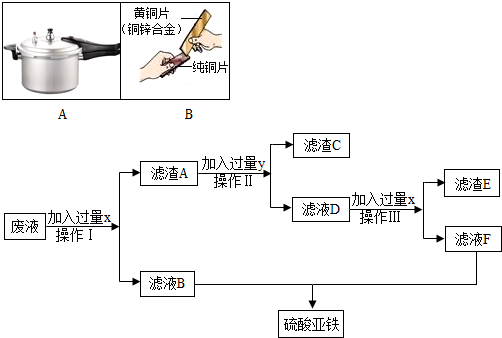
 �������������������Ӧ��ʮ�ֹ㷺��
�������������������Ӧ��ʮ�ֹ㷺�� �������������������Ӧ��ʮ�ֹ㷺��
�������������������Ӧ��ʮ�ֹ㷺��