��Ŀ����
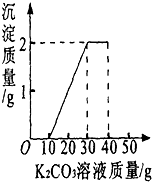
СҦͬѧ�����ȡ������̼��ʵ��Ի��յ�������Ȼ��ƻ����Һ���������������ʣ�����������ʵ�飺ȡ40g����Һ���ձ��У�����40g������������Ϊ13.8%��K2CO3��Һ������K2CO3��Һ���������ɳ��������Ĺ�ϵ��ͼ��ʾ����
��1����ȡ40g��Һ��HCl��CaCl2��������
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������
��3��ʵ���������ˣ�������Һ���ɣ��õ������������
��1����ȡ40g��Һ��HCl��CaCl2��������
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������
��3��ʵ���������ˣ�������Һ���ɣ��õ������������

��1����ͼ���е����ݿ��Կ�����10��֮ǰû�г��ֳ��������Ժ����ᷴӦ��̼�����Һ��10�ˣ��Ӽ���10g̼�����Һ��ʼ���ֳ�������30g��������䣬˵�����Ȼ��Ʒ�Ӧ��̼�����Һ��20�ˣ����ɳ�����2�ˣ�
10g̼�����Һ�к���̼��ص�����Ϊ10g��13.8%=1.38g
���̼��ط�Ӧ��������X�������Ȼ���Ϊy�����ɶ�����̼������Ϊz
K2CO3+2HCl�T2KCl+H2O+CO2��
138 73 14944
1.38gx y z
=
=
=
��ã�x=0.73g��y=1.49g��z=0.44g
���Ȼ��Ʒ�Ӧ��̼��ص�����Ϊ��20g��13.8%=2.76g
���̼��ط�Ӧ���Ȼ���Ϊa�������Ȼ���Ϊb�����ɵ�̼�������Ϊc
K2CO3+CaCl2�T2KCl+CaCO3��
138 111 149100
2.76g a b c
=
=
=
��ã�a=2.22g��b=2.98g��c=2g
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������Ϊ��
��100%=6.6%
��3����Ӧ���ձ��е����ʰ���������Ӧ�����ɵ��Ȼ��أ��Լ���Ӧʣ���̼��ص��������ʣ�1.49g+2.98g+��40g-30g����13.8%=5.85g��
�𣺣�1����ȡ40g��Һ��HCl������Ϊ0.73g��CaCl2������Ϊ1.48g��
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������Ϊ6.6%��
��3��ʵ���������ˣ�������Һ���ɣ��õ����������Ϊ5.85g��
10g̼�����Һ�к���̼��ص�����Ϊ10g��13.8%=1.38g
���̼��ط�Ӧ��������X�������Ȼ���Ϊy�����ɶ�����̼������Ϊz
K2CO3+2HCl�T2KCl+H2O+CO2��
138 73 14944
1.38gx y z
| 138 |
| 1.38g |
| 73 |
| x |
| 149 |
| y |
| 44 |
| z |
��ã�x=0.73g��y=1.49g��z=0.44g
���Ȼ��Ʒ�Ӧ��̼��ص�����Ϊ��20g��13.8%=2.76g
���̼��ط�Ӧ���Ȼ���Ϊa�������Ȼ���Ϊb�����ɵ�̼�������Ϊc
K2CO3+CaCl2�T2KCl+CaCO3��
138 111 149100
2.76g a b c
| 138 |
| 2.76g |
| 111 |
| a |
| 149 |
| b |
| 100 |
| c |
��ã�a=2.22g��b=2.98g��c=2g
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������Ϊ��
| 1.49g+2.98g |
| 30g+40g-2g-0.44g |
��3����Ӧ���ձ��е����ʰ���������Ӧ�����ɵ��Ȼ��أ��Լ���Ӧʣ���̼��ص��������ʣ�1.49g+2.98g+��40g-30g����13.8%=5.85g��
�𣺣�1����ȡ40g��Һ��HCl������Ϊ0.73g��CaCl2������Ϊ1.48g��
��2��������K2CO3������Ϊ30gʱ��������Һ�����ʵ���������Ϊ6.6%��
��3��ʵ���������ˣ�������Һ���ɣ��õ����������Ϊ5.85g��

��ϰ��ϵ�д�
�����Ŀ