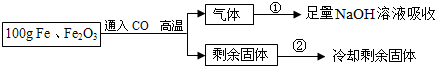
��Ŀ����
������NH3����һ����ɫ�д̼�����ζ�����壬��������ˮ������ˮ��Һ��Ϊ��ˮ���Լ��ԣ����ڻ�ѧ��ҵ��;��Ӧ�ù㷺�������ƻ��ʡ��ƴ���ȣ����������������ڻ���������
��1���������������� ��
��2���������Ƽ���ؼ�һ���ķ�Ӧԭ���ɱ�ʾΪ��NH3+CO2+H2O+NaCl=NaHCO3+A����������AҲ���������ʣ�AԪ���е�Ԫ�ص���������Ϊ ��
��3����400�����ң��ڴ������ڵ������£��ð����ɽ��ж�����NO��ԭΪN2��H2O����д���÷�Ӧ�Ļ�ѧ����ʽ ��
��1����������������
��2���������Ƽ���ؼ�һ���ķ�Ӧԭ���ɱ�ʾΪ��NH3+CO2+H2O+NaCl=NaHCO3+A����������AҲ���������ʣ�AԪ���е�Ԫ�ص���������Ϊ
��3����400�����ң��ڴ������ڵ������£��ð����ɽ��ж�����NO��ԭΪN2��H2O����д���÷�Ӧ�Ļ�ѧ����ʽ
���㣺�������ʵ����������,Ԫ�ص�������������,��ѧ�������������ʵIJ��Ӧ��,�����غ㶨�ɼ���Ӧ��,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺�������� ��ѧ����
��������1�����ʵ�����������ָ����Ҫͨ����ѧ�仯���ֳ��������ʣ���Ҫ������ɫ��״̬����ζ���ܶȡ��۵㡢�е㡢Ӳ�ȵȣ�
��2�����������غ㶨�ɷ�����
��3�����ݷ�Ӧ��������Ӧ����д����ѧ��Ӧ����ʽ��
��2�����������غ㶨�ɷ�����
��3�����ݷ�Ӧ��������Ӧ����д����ѧ��Ӧ����ʽ��
����⣺��1�����ʵ����������Dz���Ҫ������ѧ�仯�ͱ��ֳ��������ʣ��磺��ɫ��״̬����ζ���ܶȡ��ܽ��ԡ��۵㡢�е�ȣ�
�ʴ�Ϊ����ɫ�д̼�����ζ�����壬��������ˮ��
��2�����������غ㶨�ɣ�ԭ�ӵ����ࡢ��Ŀ�ڻ�ѧ��Ӧǰ���֪��AΪNH4Cl����Ԫ�ص���������Ϊ
��100%�T26.2%���ʴ�Ϊ��26.2%��
��3��������400�����ң��ڴ������ڵ������£��ð����ɽ��ж�����NO��ԭΪN2��H2O���ʴ�Ϊ��4NH3+6NO
5N2+6H2O��
�ʴ�Ϊ����ɫ�д̼�����ζ�����壬��������ˮ��
��2�����������غ㶨�ɣ�ԭ�ӵ����ࡢ��Ŀ�ڻ�ѧ��Ӧǰ���֪��AΪNH4Cl����Ԫ�ص���������Ϊ
| 14 |
| 53.5 |
��3��������400�����ң��ڴ������ڵ������£��ð����ɽ��ж�����NO��ԭΪN2��H2O���ʴ�Ϊ��4NH3+6NO
| ���� |
| 400�� |
��������Ҫ������ʶ�����ʵ��������ʣ������غ㶨�ɵ�Ӧ�ã�����ѧ����Ӧ��֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
���������еij�������һ�����л�ѧ�仯���ǣ�������
| A����դ������ |
| B�������˸ |
| C������������ |
| D������̿���� |
 �кͷ�Ӧ���ճ������ũҵ�������й㷺��Ӧ�ã�
�кͷ�Ӧ���ճ������ũҵ�������й㷺��Ӧ�ã�