��Ŀ����
21�����Ǻ��������õ����ͣ���̨�ġ�ʮ���塱�滮ȷ���ˡ���ɫ���á���չս�ԣ�����ѧ��Դ���ۺ����ý����ص㷢չ������֮һ��
��1����ˮ��������������ˮ�����ú�ˮ�и��ɷֵ� ��ͬ�������ˮ��
��2�����Σ����ƴ��Σ���±ˮ����ȡ�����õĽᾧ������ ���ڴ��δ����������к�����ɳ������þ���Ȼ��Ƶ����ʣ�����������ˮȻ��ͨ�����²������ɵõ��ϴ����Ȼ��ƣ�a���ˣ�b�ӹ�����Ba��OH��2��Һ��c�����������d�ӹ�����NaCO3��Һ��e��������ȷ�IJ���˳���� ��e ������ĸ��
��3���Ƽ識�ƴ��������ͼ��ͼ��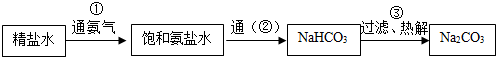
���ͼ�����ŵ����ݣ����� ��
��1����ˮ��������������ˮ�����ú�ˮ�и��ɷֵ�
��2�����Σ����ƴ��Σ���±ˮ����ȡ�����õĽᾧ������
��3���Ƽ識�ƴ��������ͼ��ͼ��

���ͼ�����ŵ����ݣ�����
���㣺�Ժ�����Դ�ĺ�������������,�ᾧ��ԭ������������Ӧ��,�Ȼ���������ᴿ,�������ȡ
ר�⣺�������� ��ѧ����
��������1����ˮ�����ķ�����Ҫ�У������ᾧ��������Ĥ�����༶���������ɸ���ԭ�����з�����
��2���ٿɸ����Ȼ����ܽ�����¶ȵĹ�ϵ���з���ѡ��ᾧ������
�ڸ��ݳ���ʱ�����������ʵ�ԭ����ʵ��Ŀ�Ľ��з�����
��3���������ʵĻ�ѧ���ʽ���ѡ���Լ���
��2���ٿɸ����Ȼ����ܽ�����¶ȵĹ�ϵ���з���ѡ��ᾧ������
�ڸ��ݳ���ʱ�����������ʵ�ԭ����ʵ��Ŀ�Ľ��з�����
��3���������ʵĻ�ѧ���ʽ���ѡ���Լ���
����⣺��1����������ˮ�����ú�ˮ�и��ɷݵķе�Ƚϸߣ�ˮ�ķе�Ƚϵͽ��з���ģ��ʴ�Ϊ���е㣻
��2����Ϊ�Ȼ��Ƶ��ܽ�����¶ȵı仯�������Բ��������ܼ����ᾧ��
Ҫ��ȥ�����к��е����ʣ����Լӹ���������������Һ��ȥ����þ���ӹ�����Na2CO3��Һ��ȥ�Ȼ��ƺ����Լ�����������ͨ�����˰Ѳ�����ˮ�����ʳ�ȥ���������������ȥ������̼���ƣ�ͨ��������ȥ���ᣮ
�ʴ�Ϊ�������ᾧ��bdace
��3����������������������з�����Ҫ�Ʊ����Ͱ�ˮ�϶��ǽ���ˮͨ��ʳ��ˮ�У��ɱ��͵İ�ˮʳ����Һ�Ʊ�̼�����ƣ��������ʵ����ʺ�Ԫ���غ�֪Ӧͨ�������̼���壻�ʴ�Ϊ��CO2
�ʴ�Ϊ����1���е㣻��2�������ᾧ��bdac����3��CO2
��2����Ϊ�Ȼ��Ƶ��ܽ�����¶ȵı仯�������Բ��������ܼ����ᾧ��
Ҫ��ȥ�����к��е����ʣ����Լӹ���������������Һ��ȥ����þ���ӹ�����Na2CO3��Һ��ȥ�Ȼ��ƺ����Լ�����������ͨ�����˰Ѳ�����ˮ�����ʳ�ȥ���������������ȥ������̼���ƣ�ͨ��������ȥ���ᣮ
�ʴ�Ϊ�������ᾧ��bdace
��3����������������������з�����Ҫ�Ʊ����Ͱ�ˮ�϶��ǽ���ˮͨ��ʳ��ˮ�У��ɱ��͵İ�ˮʳ����Һ�Ʊ�̼�����ƣ��������ʵ����ʺ�Ԫ���غ�֪Ӧͨ�������̼���壻�ʴ�Ϊ��CO2
�ʴ�Ϊ����1���е㣻��2�������ᾧ��bdac����3��CO2
�����������ѶȱȽϴ��漰��ˮ����������ԭ������ˮɹ�ε�ԭ�������ӵ�ԭ���������Ƽ��ԭ�����������ݣ�Ҫͨ�����������ܽᣮ

��ϰ��ϵ�д�
 ����������������ϵ�д�
����������������ϵ�д�
�����Ŀ
���ڻ�ѧ��Ӧ����ʽS+O2
SO2��ʾ�����壬����������ǣ�������
| ||
| A��ÿ32�����������32��������������ȫ��Ӧ������64�������Ķ������� |
| B�����������Ӧ���ɶ������� |
| C����������ڵ�ȼ�������������˶������� |
| D���ڵ�ȼ�������£�ÿһ����ԭ�Ӻ�һ�������ӻ�������һ������������� |
���������У������ù���NaOH������ǣ�������
| A��O2 |
| B��CO2 |
| C��H2 |
| D��CO |
���������У���Ԫ��������̬���ڵ��ǣ�������
| A��H2O2 |
| B��H3PO4 |
| C��O3 |
| D��HgO |
ʳƷ��ȫ��ϵ��ǧ����ÿ���˵����彡����ʳ������ʳ���Ӱ�����彡�����ǣ�������
| A����С�մͷ۱��Ƶĸ�� |
| B���ü�ȩ��Һ���ݵ�ˮ��Ʒ |
| C�����������͵�ζ���������� |
| D���üӸ��Ȼ������Ƶ� ���� |

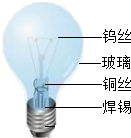 �����ͽ����������������������Ź㷺��Ӧ�ã���ش��������⣺
�����ͽ����������������������Ź㷺��Ӧ�ã���ش��������⣺