��Ŀ����
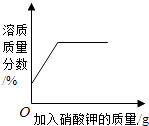
����Ŀ�����������еij���Ԫ�أ�ȱ��ʱ��ͨ��ʳ�ñ���ҩ����������������ij����ҩ��˵����IJ�����Ϣ��ͼ��ʾ���ֽ�100g����ֳ�5�ȷݣ���μӵ���40Ƭ��ҩ���Ƴɵķ�ĩ�У������ɷֲ������ᷴӦ�����õ�������������ͼ2��������й���Ϣ�ش����⣮ 
��1�� | ��3�� | ��4�� | |
���������������g�� | 20 | 20 | 20 |
ʣ������������g�� | 35 | a | 20 |
��1������ȱ���׳��ֵļ����� �� ��ʱ����ҽ��ָ���·��ñ���ҩ���⣬�ճ�������Ҫ��������ʳ�
��2����Ʒ�Ʋ���ҩ����CaCO3������������ �� a����ֵΪ ��
��3��������������ʵ���������Ϊ��д��������̣�����������һλС������
���𰸡�
��1���������ɣ�ϺƤ����ţ�̻��ͷ����
��2��50%��25
��3���⣺�ɱ�����Ϣ��֪��80gϡ������40Ƭҩ���е�̼���ǡ����ȫ��Ӧ�����Ը����������ʵ���������Ϊ�� ![]() =18.3%��
=18.3%��
�𣺷�Ӧ�����������ʵ�����������18.3%��
���������⣺��1������ȱ���׳��ֹ������ɣ��ճ�������Ҫ������ϺƤ����ţ�̻��ͷ���Ⱥ��н϶�ĸ�Ԫ�ص�ʳ���2����40Ƭ��ҩ���к�̼��Ƶ�����Ϊx�����������е���������Ϊy��
CaCO3+ | 2HCl�T | CaCl2+H2O+ | CO2�� |
100 | 73 | 44 | |
x | y | 8.8g |
![]() ��
�� ![]()
x=20g��y=14.6g
��Ʒ�Ʋ���ҩ����CaCO3�����������ǣ� ![]() =50%��
=50%��
�ɵ�1�κ͵�4�����ݿ�֪20gϡ���������5g̼��ƣ���ڶ��μ���20gϡ�����ʣ�����Ϊ30g�������μ���30gϡ�����ʣ�����Ϊ25g����a����ֵΪ25��
�𰸣���1���������ɣ�ϺƤ����ţ�̻��ͷ���ȣ���2��50%��25��
�����㾫�������ø��ݻ�ѧ��Ӧ����ʽ�ļ������Ŀ�����жϼ��ɵõ��𰸣���Ҫ��֪�����ʼ�������=ϵ������Է�������֮�ȣ�

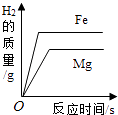
����Ŀ������ͼ������ȷ��ӳ��Ӧ�仯��ϵ���ǣ� ��
|
|
|
|
A.20��ʱ����һ����������ز�������Һ�м�������ع��� | B������������þƬ����ƬͶ�뵽����ϡ������ | C���þƾ��Ƽ���һ�����ĸ�����ع��� | D����ʢ�������������̵��ձ��в��ϵؼ������������Һ |
A.A
B.B
C.C
D.D
����Ŀ��M�������г����Ľ�����MSO4��Һ����ɫҺ�壬ij��ѧ��ȤС���ͬѧ��̽��M��ͭ�����Ļ��˳�����������һ���������̽�����
��1����������⡿M��ͭ�����Ľ������˳���������ģ� ����������衿С���IJ�����Al��Cu��M��Сǿ�IJ�����Al��M��Cu����IJ����� ��
��2���������ʵ��һ��С��ȡM˿��ͭ˿����˿�ͱ��Ϊ�١��ڡ��۵�3֧�Թܣ����������ʵ�飨�Թ��о�ʢ�������ͬ��������������Ҳ��ͬ��ϡ���ᣩ��
ʵ����� | M˿�����Թܢ��� | ͭ˿�����Թܢ��� | ��˿�����Թܢ��� | ʵ����� |
�տ�ʼ������ | ���������ݲ��� | ������ | ������ | M��Cu�� |
�����Ӻ������ | ���������ݲ��� | ������ | �д������ݲ��� |
��3������������ͬѧ�Ƕ�ͬһʵ��ó����ֲ�ͬ���۵�������������ۣ�������ʵ��տ�ʼʱ�Թܢ��������ݲ�����ԭ��ԭ���� ��
��4���������ʵ�����Ϊ�˽�һ��̽�����ֽ����Ļ��˳��ͬѧ��ȡ��ϸ��ͬ����ĥ�ɾ���M˿����˿�������Һ���ֽ�������ͼ��ʾ��ʵ�飺 
���Թ����й۲쵽�������� ��
���Թܱ��з�Ӧ�Ļ�ѧ����ʽΪ ��
��5������������ͬѧ�Ǿ����ۺ���Ϊ��ʵ������Ż���ȥ���Թ������Թܱ�ţ��е�ʵ�飬Ҳ���Եó���ȷ�Ľ��ۣ� ����������ۡ�ͨ������ʵ�飬ͬѧ�ǵó������ֽ����Ļ��˳��
��6���������뷴˼��ʵ�������ͬѧ�Ƕ�ʵ����̽��������·�˼�� ��Ӱ��������ᷴӦ���ʿ��������أ��������Ӧ�Ľ�������������⣬��������дһ�֣�
��̽���������˳��ʱ����������ʵ�����Ѿ��õ������ַ����⣬�����Ը��������жϣ���дһ�֣�
����Ŀ��ijС��ѧϰ��������̼��ȡ���о�������ʱ��̽���˶�����̼������ռ�������
���������ϡ�
ͨ��״���£�1 ���ˮԼ���ܽ�1 ���������̼��������ҺPH ԼΪ5.6��
��������⡿
������̼�ܲ�������ˮ���ռ���
��ʵ������������
��1����ͨ��״���£��ⶨ������̼��Һˮ������Һ����PH���ж϶�����̼��ˮ���ܽ�ȵ������
��װ���з�Ӧ�Ļ�ѧ����ʽ����
��2��ʵ��ʱ�����Ƚ���װ���еĿ����ž���������ǣ��������ɼ�������K1��K2��K3 �� ��ͬ���رյ��ɼ� �� �������μ�ϡ�����������ž�������������ž��ķ�������
��3���ر�K1 �� ��K2��K3������װ�����ռ���ƿ����ʱ���رջ���K2 ��K3 �� �����װ�á�Ȼ����pH�Ʋ���������ݣ�
���� | ��װ������Һ | ��װ������Һ |
pH | 6.50 | 5.60 |
������֪���ڱ��Ͷ�װ����ˮ���ܽ������̼�����������ڡ�����С�ڡ��������ڡ��� ���Ͷ�װ������Һ���������
��4��ʵ���У���װ�õ�����������û����װ�ã���������ҺpH ����
��5����ʵ�����������Ϊ������̼����ܡ����ܡ�������ˮ���ռ����ܼ��ٶ�����̼��ˮ���ܽ�Ĵ�ʩ������дһ�����ɣ�