��Ŀ����
��15�֣�ijͬѧ���ʯ��ȼ�տ�չ��������̽��������������̽�����̵�������⣺
��������⡿ʯ���г���̼����Ԫ���⣬�Ƿ�����Ԫ�أ�
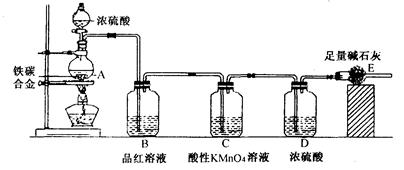
��ʵ����ơ����������غ㶨�ɣ���ͬѧ�þ���ʯ����������о�����������������ʵ�飨����Ũ���ᡢ��ˮ����ͭ��������ˮ�֣�ҩƷ������֤���㣬���̶ֹ�װ��ʡ�ԣ���

������ԭ����
��1����д��Aװ�ã��������ܣ������Եļ�鷽���� ��װ��A�з�����Ӧ�Ļ�ѧ����ʽ�� ��
��2��װ��B�������� ��װ��D�������� ��
��3��װ��F������Ӧ�Ļ�ѧ����ʽ�� ��
��4��Ϊ�ﵽʵ��Ŀ�ģ���Ӧǰ���������� ����װ����ţ�װ���е�ҩƷ������
�����ݴ�����
��5����ͬѧ����ʦ�����£�����һЩ����ʵ���������ó����������ݣ��������������ݷ��������۲��֡�
�����۽������������봦����
��6�����ݸ�ѧ����Ƶ�ʵ�飬����Ϊ�������ԭ���� ����дһ������
��7���������������ԭ�������������ʵ���Ľ���ʩ�� ��
��������⡿ʯ���г���̼����Ԫ���⣬�Ƿ�����Ԫ�أ�
��ʵ����ơ����������غ㶨�ɣ���ͬѧ�þ���ʯ����������о�����������������ʵ�飨����Ũ���ᡢ��ˮ����ͭ��������ˮ�֣�ҩƷ������֤���㣬���̶ֹ�װ��ʡ�ԣ���

������ԭ����
��1����д��Aװ�ã��������ܣ������Եļ�鷽���� ��װ��A�з�����Ӧ�Ļ�ѧ����ʽ�� ��
��2��װ��B�������� ��װ��D�������� ��
��3��װ��F������Ӧ�Ļ�ѧ����ʽ�� ��
��4��Ϊ�ﵽʵ��Ŀ�ģ���Ӧǰ���������� ����װ����ţ�װ���е�ҩƷ������
�����ݴ�����
��5����ͬѧ����ʦ�����£�����һЩ����ʵ���������ó����������ݣ��������������ݷ��������۲��֡�
| ʵ����ʵ | ���ݷ��������� |
| 2.12 gʯ����ȫȼ�գ��õ�6.6g CO2��2.88g H2O | д��������̣� ���ۣ� �� |
��6�����ݸ�ѧ����Ƶ�ʵ�飬����Ϊ�������ԭ���� ����дһ������
��7���������������ԭ�������������ʵ���Ľ���ʩ�� ��
��1���Ƚ����ܷ���ˮ�У�������Һ©����رշ�Һ©�����ţ�������ס��ƿ�ײ��������ܿ�������ð��������Aװ�����������ã�H2O2 H2O+O2��
H2O+O2��
��2������������ȷ������ȫȼ�յ�һ����̼��̼ȫ��ת��Ϊ������̼
��3��2NaOH+CO2=Na2CO3+H2O
��4��C��E��F
��5����2�֣����ݷ���m(C)=6.6g��12/44=1.8g m(H)=2.88g��2/18=0.32g
m(C)+m(H)=1.8g+0.32g=2.12g=m��ʯ����
��1�֣����ۣ�ʯ���в�����Ԫ��
��6��ԭ��װ���ڲ����Ŀ�������ˮ�Ͷ�����̼��������оȼ��Ҳ�����ˮ�Ͷ�����̼��δ��ȫȼ�յ�ʯ��������̼�ڲ����ڵ����ڻ�δ���ж���ظ�ʵ���
��7����ʩ��ʵ��ǰ��ͨ������װ���ڵĿ����Ͼ���ȡ��ͬ����������о������������ͬ��ʵ�飬Ȼ��۳���Ӧ�����ݻ���ж���ظ�ʵ��ȡƽ��ֵ��
 H2O+O2��
H2O+O2����2������������ȷ������ȫȼ�յ�һ����̼��̼ȫ��ת��Ϊ������̼
��3��2NaOH+CO2=Na2CO3+H2O
��4��C��E��F
��5����2�֣����ݷ���m(C)=6.6g��12/44=1.8g m(H)=2.88g��2/18=0.32g
m(C)+m(H)=1.8g+0.32g=2.12g=m��ʯ����
��1�֣����ۣ�ʯ���в�����Ԫ��
��6��ԭ��װ���ڲ����Ŀ�������ˮ�Ͷ�����̼��������оȼ��Ҳ�����ˮ�Ͷ�����̼��δ��ȫȼ�յ�ʯ��������̼�ڲ����ڵ����ڻ�δ���ж���ظ�ʵ���
��7����ʩ��ʵ��ǰ��ͨ������װ���ڵĿ����Ͼ���ȡ��ͬ����������о������������ͬ��ʵ�飬Ȼ��۳���Ӧ�����ݻ���ж���ظ�ʵ��ȡƽ��ֵ��
��1����װ��һ���Dz���Һ�ⷨ���������Եļ��飮��2��������������ˮ�ԣ�����ͭ��һ����̼��̼���ɶ�����̼��ͭ���н��3���������ƺͶ�����̼����̼���ƺ�ˮ
��4�����������غ㶨�ɣ��ڻ�ѧ��Ӧ�У��μӷ�Ӧǰ�����ʵ������ܺ͵��ڷ�Ӧ�����ɸ����ʵ������ܺͣ����н��
��5�����ݶ�����̼��̼��������ˮ���������֮����ʯ�����������бȽϣ��������
��6��ԭ��װ���ڲ����Ŀ�������ˮ�Ͷ�����̼��������оȼ��Ҳ�����ˮ�Ͷ�����̼��δ��ȫȼ�յ�ʯ��������̼�ڲ����ڵ����ڻ�δ���ж���ظ�ʵ���
��7����ʩ��ʵ��ǰ��ͨ������װ���ڵĿ����Ͼ���ȡ��ͬ����������о������������ͬ��ʵ�飬Ȼ��۳���Ӧ�����ݻ���ж���ظ�ʵ��ȡƽ��ֵ��
��4�����������غ㶨�ɣ��ڻ�ѧ��Ӧ�У��μӷ�Ӧǰ�����ʵ������ܺ͵��ڷ�Ӧ�����ɸ����ʵ������ܺͣ����н��
��5�����ݶ�����̼��̼��������ˮ���������֮����ʯ�����������бȽϣ��������
��6��ԭ��װ���ڲ����Ŀ�������ˮ�Ͷ�����̼��������оȼ��Ҳ�����ˮ�Ͷ�����̼��δ��ȫȼ�յ�ʯ��������̼�ڲ����ڵ����ڻ�δ���ж���ظ�ʵ���
��7����ʩ��ʵ��ǰ��ͨ������װ���ڵĿ����Ͼ���ȡ��ͬ����������о������������ͬ��ʵ�飬Ȼ��۳���Ӧ�����ݻ���ж���ظ�ʵ��ȡƽ��ֵ��

��ϰ��ϵ�д�
�����Ŀ