摘要:下列离子方程式书写正确的是 A.Ca(ClO)2溶液中通入过量CO2:Ca2++2C1O-+H2O+CO2 CaCO3↓+2HC1O B.Ba(OH)2溶液中滴入NaHSO4溶液至恰好为中性: 2H++SO42-+Ba2++2OH- BaSO4↓+2H2O C.金属钠投入水中:2Na+H2O===2Na++OH-+H2↑ D.NaOH溶液中通入C12:2C12+2OH- 3C1-+C1O-+H2O
网址:http://m.1010jiajiao.com/timu3_id_425620[举报]
下列离子方程式书写正确的是
A.Ca(ClO)2溶液中通入过量CO2:Ca2++2C1O-+H2O+CO2 CaCO3↓+2HC1O
B.Ba(OH)2溶液中滴入NaHSO4溶液至恰好为中性:
2H++SO42-+Ba2++2OH-
BaSO4↓+2H2O
C.金属钠投入水中:2Na+H2O===2Na++OH-+H2↑
D.NaOH溶液中通入C12:2C12+2OH- 3C1-+C1O-+H2O
查看习题详情和答案>>
下列离子方程式书写正确的是
- A.Ca(HCO3)2溶液中滴入过量NaOH溶液:HCO
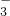 +Ca2++OH-=CaCO3↓+H2O
+Ca2++OH-=CaCO3↓+H2O - B.向NaAlO2溶液中通入过量的CO2:CO2+2H2O+AlO2
 =Al(OH)3↓+HCO
=Al(OH)3↓+HCO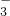
- C.0.01mol/L NH4Al(SO4)2溶液与0.02mol/L Ba(OH)2溶液等体积混合:Al3++2SO42-+2Ba2++4OH-=2BaSO4↓+AlO
 +2H2O
+2H2O - D.向Ca(ClO)2溶液中通入过量的SO2:ClO-+SO2+H2O=HClO+HSO
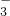
下列离子方程式书写正确的是
A.Ca(ClO)2溶液中通入过量CO2:Ca2++2C1O-+H2O+CO2 CaCO3↓+2HC1O
B.Ba(OH)2溶液中滴入NaHSO4溶液至恰好为中性:
2H++SO42-+Ba2++2OH- BaSO4↓+2H2O
C.金属钠投入水中:2Na+H2O===2Na++OH-+H2↑
D.NaOH溶液中通入C12:2C12+2OH- 3C1-+C1O-+H2O