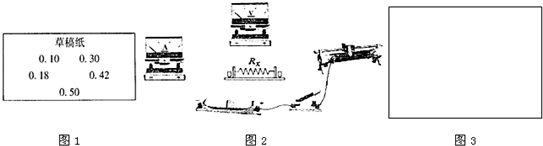
��Ŀ����
�ڲ���ijһ��ֵ����Rx��ֵ��ʵ���У����ͬѧѡ������ͼ��ʾ�����Ľ���ʵ�飮ʵ������У���������ͬѧ�����������ʵ������ŷ��֣��Ѳ����ĵ���ֵ��д�ڲݸ�ֽ�϶��������˼�¼�����С�
ʵ����� | 1 | 2 | 3 | 4 | 5 |
ѹU��V | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 |
����I��A |
��1�������������ͬѧ�Ѽ�¼�ڲݸ�ֽ�ϵĵ���ֵ��Ӧ���˱����У���д���������ݵ����������� �������������������������������������������������������������� ��2�֣�
��2�����ݲ������ݼ���ö�ֵ�������ֵΪ �������������� ��������һλС������2�֣�
��3������ʵ�������·���ñʻ��ߴ��浼���������ͼ�е�ʵ�����ӡ���2�֣�
��4��������������ʵ���г����𱣻���·�������⣬��һ����Ҫ��������������������
���������������������������������������������������������������� ����2�֣�

![]() ��5������Դ��ѹ�㶨��δ֪���õ�����R����·���š����� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
��5������Դ��ѹ�㶨��δ֪���õ�����R����·���š����� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�


��10�֣��ڲ���ijһ��ֵ����Rx��ֵ��ʵ���У����ͬѧѡ������ͼ��ʾ�����Ľ���ʵ�飮ʵ������У���������ͬѧ�����������ʵ������ŷ��֣��Ѳ����ĵ���ֵ��д�ڲݸ�ֽ�϶��������˼�¼�����С�
| ʵ����� | 1 | 2 | 3 | 4 | 5 |
| ѹU��V | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 |
| ����I��A | | | | | |

��1�������������ͬѧ�Ѽ�¼�ڲݸ�ֽ�ϵĵ���ֵ��Ӧ���˱����У���д���������ݵ����������� ��2�֣�
��2�����ݲ������ݼ���ö�ֵ�������ֵΪ ��������һλС������2�֣�
��3������ʵ�������·���ñʻ��ߴ��浼���������ͼ�е�ʵ�����ӡ���2�֣�


��4��������������ʵ���г����𱣻���·�������⣬��һ����Ҫ������
����2�֣�
 ��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣� ��10�֣��ڲ���ijһ��ֵ����Rx��ֵ��ʵ���У����ͬѧѡ������ͼ��ʾ�����Ľ���ʵ�飮ʵ������У���������ͬѧ�����������ʵ������ŷ��֣��Ѳ����ĵ���ֵ��д�ڲݸ�ֽ�϶��������˼�¼�����С�
|
ʵ����� |
1 |
2 |
3 |
4 |
5 |
|
ѹU��V |
0.5 |
1.0 |
1.5 |
2.0 |
2.5 |
|
����I��A |
|
|
|
|
|

��1�������������ͬѧ�Ѽ�¼�ڲݸ�ֽ�ϵĵ���ֵ��Ӧ���˱����У���д���������ݵ����������� ��2�֣�
��2�����ݲ������ݼ���ö�ֵ�������ֵΪ ��������һλС������2�֣�
��3������ʵ�������·���ñʻ��ߴ��浼���������ͼ�е�ʵ�����ӡ���2�֣�


��4��������������ʵ���г����𱣻���·�������⣬��һ����Ҫ������
����2�֣�
 ��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
�ڲ���ijһ��ֵ����Rx��ֵ��ʵ���У����ͬѧѡ������ͼ��ʾ�����Ľ���ʵ�飮ʵ������У���������ͬѧ�����������ʵ������ŷ��֣��Ѳ����ĵ���ֵ��д�ڲݸ�ֽ�϶��������˼�¼�����С�
| ʵ����� | 1 | 2 | 3 | 4 | 5 |
| ѹU��V | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 |
| ����I��A |

��1�������������ͬѧ�Ѽ�¼�ڲݸ�ֽ�ϵĵ���ֵ��Ӧ���˱����У���д���������ݵ����������� ��2�֣�
��2�����ݲ������ݼ���ö�ֵ�������ֵΪ ��������һλС������2�֣�
��3������ʵ�������·���ñʻ��ߴ��浼���������ͼ�е�ʵ�����ӡ���2�֣�
 | |||
 | |||
��4��������������ʵ���г����𱣻���·�������⣬��һ����Ҫ������
����2�֣�
![]() ��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
��5������Դ��ѹ�㶨��δ֪���õ�����R����·���� �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ����������ͼ�еķ����л�����2�֣�
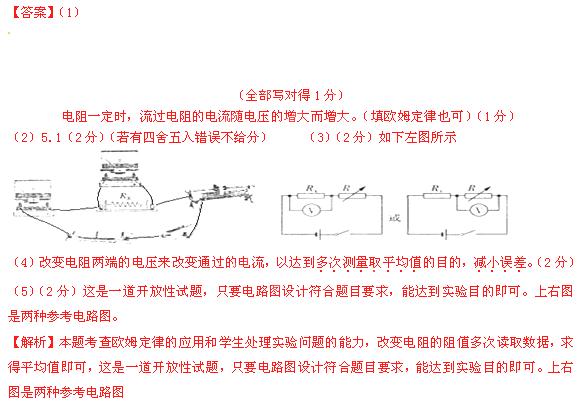


 �����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ������ͼ3�еķ����л�����2�֣�
�����滬����������ͬʱ��ȥ���������������������ģ���Ҫ�����������κ����ģ����һ������RX��ֵ��ʵ���·ͼ������ͼ3�еķ����л�����2�֣�