جâؤ؟ؤعبف
°´زھاَذ´³ِ298K،¢101 kPaت±دآءذ·´س¦µؤبب»¯ر§·½³جت½£؛
£¨1£©3mol NO2(g)سë1 mol H2O(1)·´س¦ةْ³ةHNO3(aq)؛حNO(g)£¬·إبب138kJ£؛____________،£
£¨2£©1 mol HgO(s)·ض½âخھز؛ج¬¹¯؛حرُئّ£¬خüبب90.7kJ£؛___________________،£
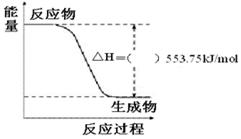
£¨3£©¢ظدآح¼تاز»¶¨ء؟±ûحéحêب«ب¼ةصةْ³ةCO2؛ح1 mol H2O(l)¹³جضذµؤؤـء؟±ن»¯ح¼£¬اëشع¸½ح¼ضذµؤہ¨؛إؤعجîبë،°£«،±»ٍ،°،ھ،±،£
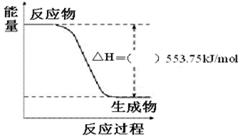
¢عذ´³ِ±يت¾±ûحéب¼ةصببµؤبب»¯ر§·½³جت½£؛_______________________________________،£
£¨1£©3NO2 (g) + H2O (l) = 2HNO3 (aq) + NO(g) ![]() H = ¨C138kJ،¤mol¨C1،£
H = ¨C138kJ،¤mol¨C1،£
£¨2£©2HgO (s) = 2Hg (l) + O2 (g) ![]() H = +181.4kJ،¤mol¨C1،£
H = +181.4kJ،¤mol¨C1،£
£¨3£©¢ظ،°،ھ،±£»¢عC3H8(g)£«5O2(g)،ْ3CO2(g)£«4H2O(l) ??H£½£2215 kJ/mol ،£

ء·د°²لدµءذ´ً°¸
 شؤ¶ء؟ى³µدµءذ´ً°¸
شؤ¶ء؟ى³µدµءذ´ً°¸
دà¹طجâؤ؟