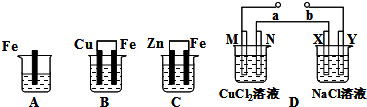
��Ŀ����
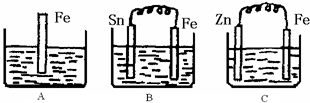 A��B��C�����ձ��зֱ�ʢ��200mL��ͬ���ʵ���Ũ�ȵ�ϡ���
A��B��C�����ձ��зֱ�ʢ��200mL��ͬ���ʵ���Ũ�ȵ�ϡ�����1���ֱ�д������װ������Ƭ���淢����Ӧ�����ӷ���ʽ��
A
Fe+2H+=Fe2++H2��
Fe+2H+=Fe2++H2��
��BFe-2e-=Fe2+
Fe-2e-=Fe2+
�� C2H++2e-=H2��
2H++2e-=H2��
����2��һ��ʱ���B��Sn��������������Һ��pH
����
����
�����������С���������䡱������3��һ��ʱ���C�в�����3.36L����״��������ʱ������ǡ��ȫ�������ģ���ԭϡ������Һ�����ʵ���Ũ��Ϊ
0.75
0.75
mol?L-1����������1���������������ܷ����û���Ӧ��Sn��Fe�����ṹ�ɵ�ԭ����У��������ϻ��ã���������Zn��Fe�����ṹ�ɵ�ԭ����У�����п�ϻ��ã���������
��2��ԭ��ص������ǵ���ʵ������ӷ����õ��ӵĻ�ԭ��Ӧ�����������ӵ����ı仯�жϣ�
��3�����������ӵķŵ練Ӧ�Լ������Ӻ����������֮�����Ĺ�ϵ�����㣮
��2��ԭ��ص������ǵ���ʵ������ӷ����õ��ӵĻ�ԭ��Ӧ�����������ӵ����ı仯�жϣ�
��3�����������ӵķŵ練Ӧ�Լ������Ӻ����������֮�����Ĺ�ϵ�����㣮
����⣺��1��A���ǽ������������ܷ����û���Ӧ��ԭ������ʽΪ��Fe+2H+=Fe2++H2����B��Sn��Fe�����ṹ�ɵ�ԭ��أ��������ϻ��ã����������缫��ӦΪ��Fe-2e-=Fe2+��C�У�Zn��Fe�����ṹ�ɵ�ԭ��أ�����п�ϻ��ã������������������������ü������ĵ缫��ӦΪ��2H++2e-=H2�����ʴ�Ϊ��Fe+2H+=Fe2++H2����Fe-2e-=Fe2+��2H++2e-=H2����
��2��B��Sn�������������ĵ缫��ӦΪ��2H++2e-=H2��������������Ũ�ȼ�С�����Լ�����pH���ʴ�Ϊ������
��3��C�����������ĵ缫��ӦΪ��2H++2e-=H2����������3.36L����״������0.15mol����ʱ�����������ӵ����ʵ���Ϊ0.3mol������ԭϡ������Һ�������ӵ����ʵ���Ϊ0.3mol��������������ʵ���0.15mol��c=
=
=0.75mol/L���ʴ�Ϊ��0.75��
��2��B��Sn�������������ĵ缫��ӦΪ��2H++2e-=H2��������������Ũ�ȼ�С�����Լ�����pH���ʴ�Ϊ������
��3��C�����������ĵ缫��ӦΪ��2H++2e-=H2����������3.36L����״������0.15mol����ʱ�����������ӵ����ʵ���Ϊ0.3mol������ԭϡ������Һ�������ӵ����ʵ���Ϊ0.3mol��������������ʵ���0.15mol��c=
| n |
| V |
| 0.15mol |
| 0.2L |
���������⿼��ԭ��صĸ������ԭ��֪ʶ�����Ը�����ѧ֪ʶ���лش��ѶȲ���

��ϰ��ϵ�д�
 ����������ϵ�д�
����������ϵ�д�
�����Ŀ
|
Ũ����ͬ��NaOH��Һ��100 ml����A��B��C�����ձ��У��ֱ���������Һ��ͨ�벻������CO2����ַ�Ӧ����������������Һ����μ���0.2 mol/L�����ᣬ����CO2������������������֮���ϵ����ͼ��ʾ�������ж���ȷ����
| |
A�� |
ԭNaOH��Һ��Ũ��Ϊ0.2 mol/L |
B�� |
B�ձ���ͨ���CO2���Ϊ448 mL |
C�� |
ͨ��CO2����ȷ��A�ձ��е����� |
D�� |
ͨ��CO2��C�ձ������ʳɷֵ����ʵ���֮��Ϊ n(NaOH)��n(Na2CO3)��2��1 |