��Ŀ����
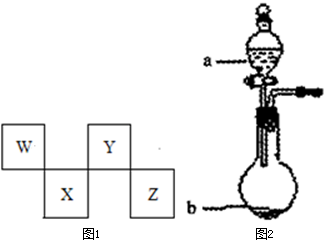
W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е�λ����ͼ��ʾ������Zλ�ڢ�A�塣��ش��������⡣

��1��W��ԭ�ӽṹʾ��ͼΪ__________����˵��Z�ķǽ����Ա�X�Ľ�����ǿ����ʵ��____________����һ������
��2��W��Y������γ�ԭ�Ӹ�����Ϊ1:1:4�Ļ������ҵ�ƾ��к��д����ʡ��û������һ����Ҫ����������ȼ�ϵ�أ���ͼ��2004�������������Ը�����Ϊȼ�ϵ�ȼ�ϵ�ؽṹʾ��ͼ����aͨ�������Ϊ
____________��B�缫��ӦʽΪ____________________��
��2��W��Y������γ�ԭ�Ӹ�����Ϊ1:1:4�Ļ������ҵ�ƾ��к��д����ʡ��û������һ����Ҫ����������ȼ�ϵ�أ���ͼ��2004�������������Ը�����Ϊȼ�ϵ�ȼ�ϵ�ؽṹʾ��ͼ����aͨ�������Ϊ
____________��B�缫��ӦʽΪ____________________��

��3�����NaZˮ��Һʱ�������ĵ缫��ӦʽΪ________________________����ҵ�ϣ���������Ӧ�����������Ca(OH)2Ϊԭ�ϣ��Ʊ�Ư�۷�Ӧ�Ļ�ѧ����ʽΪ________________________��
��4����֪��298Kʱ����������Y2���巴Ӧ������1 mol Na2Y����ʱ���ų�����414 kJ������1 mol Na2Y2
����ʱ���ų�����511 kJ����Na2Y������Y2���巴Ӧ����Na2Y2������Ȼ�ѧ����ʽΪ________________________��
��4����֪��298Kʱ����������Y2���巴Ӧ������1 mol Na2Y����ʱ���ų�����414 kJ������1 mol Na2Y2
����ʱ���ų�����511 kJ����Na2Y������Y2���巴Ӧ����Na2Y2������Ȼ�ѧ����ʽΪ________________________��
��1�� �������������ǿ������ ����HCl���ȶ���ǿ��PH3��
�������������ǿ������ ����HCl���ȶ���ǿ��PH3��
��2��CH3OH��O2+4H++4e-=2H2O
��3��2H++2e-=H2��2Cl2+2Ca(OH)2==CaCl2+Ca(ClO)2+2H2O
��4��Na2O(s)+1/2O2(g)=Na2O2(s) ��H=-97kJ/mol
 �������������ǿ������ ����HCl���ȶ���ǿ��PH3��
�������������ǿ������ ����HCl���ȶ���ǿ��PH3�� ��2��CH3OH��O2+4H++4e-=2H2O
��3��2H++2e-=H2��2Cl2+2Ca(OH)2==CaCl2+Ca(ClO)2+2H2O
��4��Na2O(s)+1/2O2(g)=Na2O2(s) ��H=-97kJ/mol

��ϰ��ϵ�д�
�����Ŀ
W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е����λ����ͼ��ʾ��������������֮�͵���24�������жϲ���ȷ���ǣ�������
| W | X | |
| Y | Z |
| A��ԭ�Ӱ뾶��С��W��X |
| B��Ԫ��������ۣ�W��Z |
| C���������ӵĻ�ԭ�ԣ�Y��Z |
| D����̬�⻯����ȶ��ԣ�X��Y |
 W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е�λ����ͼ��ʾ������Zλ�ڢ�A�壮��ش��������⣮
W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е�λ����ͼ��ʾ������Zλ�ڢ�A�壮��ش��������⣮