��Ŀ����
��һ����ɫ���壨Ϊһ�ֶ�Ԫ������Σ����±�Ϊ�������ʵ��IJ���ʵ�鱨�森
��1�������ʵ���������������ж����ڱ����ڵ���Ӧλ�ã�
��2�������һ��ȷ�ϴ˰�ɫ����ijɷ֣�Ӧ�ڲ���
��3���������Լ�A�����а�ɫ������д���ݴ�ȷ�ϵ���ҺX������������Һ���ȷ�Ӧ�Ļ�ѧ����ʽ
��1�������ʵ���������������ж����ڱ����ڵ���Ӧλ�ã�
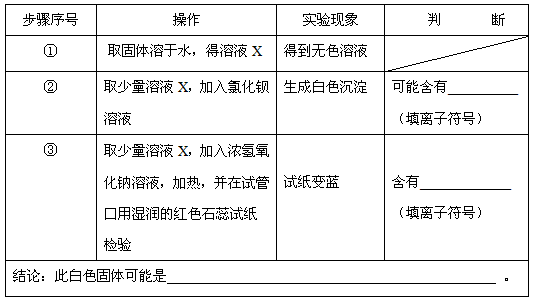
| ������� | ���� | ʵ������ | �� �� |
| �� | ȡ��������ˮ������ҺX | �õ���ɫ��Һ | |
| �� | ȡ������ҺX�������Ȼ�����Һ | ���ɰ�ɫ���� | ���ܺ��� SO42-��SO32-��CO32-��δдSO32-���۷֣� SO42-��SO32-��CO32-��δдSO32-���۷֣� ����д���֣������ӷ��ţ� |
| �� | ȡ������ҺX������Ũ����������Һ�����ȣ������Թܿ���ʪ��ĺ�ɫʯ����ֽ���� | ��ֽ���� | ���� NH4+ NH4+ �������ӷ��ţ� |
| ���ۣ��˰�ɫ��������� ��NH4��2SO4��NH4��2SO3��NH4��2CO3��δд��NH4��2SO3���۷֣� ��NH4��2SO4��NH4��2SO3��NH4��2CO3��δд��NH4��2SO3���۷֣� �� | |||
��
��
�������ţ��м����Լ�A���Լ�AΪ����
����
����3���������Լ�A�����а�ɫ������д���ݴ�ȷ�ϵ���ҺX������������Һ���ȷ�Ӧ�Ļ�ѧ����ʽ
2NH4++SO42-+Ba2++2OH-
BaSO4��+2NH3��+2H2O
| ||
2NH4++SO42-+Ba2++2OH-
BaSO4��+2NH3��+2H2O
��
| ||
��������1���ڼ����Ȼ�����Һ���ɳ��������ܺ���SO42-��SO32-��CO32-���ӣ�
�۸�������ӵļ��鷽�������жϣ�
�������Ϸ��������Ʋ����ɫ����ɷ֣�
��2���ڢ��м������ᣬ���������ж�������ɣ�
��3������������Ȼ���ɳ���������Ϊ���ᱵ����ɫ����Ϊ����泥�
�۸�������ӵļ��鷽�������жϣ�
�������Ϸ��������Ʋ����ɫ����ɷ֣�
��2���ڢ��м������ᣬ���������ж�������ɣ�
��3������������Ȼ���ɳ���������Ϊ���ᱵ����ɫ����Ϊ����泥�
����⣺��1�������ڼ����Ȼ�����Һ���ɳ��������ܺ���SO42-��SO32-��CO32-���ӣ�
�ʵ�λ��SO42-��SO32-��CO32-��
�ۼ���Ũ����������Һ�����ȣ������Թܿ���ʪ��ĺ�ɫʯ����ֽ���飬��ֽ������֤����������ӣ�
�ʴ�Ϊ��NH4+��
�������Ϸ�������ɫ�������Ϊ������NH4��2SO4��NH4��2SO3��NH4��2CO3
�ʴ�Ϊ����NH4��2SO4��NH4��2SO3��NH4��2CO3��
��2�����һ��ȷ�ϴ˰�ɫ����ijɷ֣���Ҫ�ڲ���ڼ���������м��飬
�ʴ�Ϊ���ڣ����
��3�����ڼ���������Ȼ���ɳ���������Ϊ���ᱵ����ɫ����Ϊ����泥���Ӧ�����ӷ���ʽΪ��2NH4++SO42-+Ba2++2OH-
BaSO4��+2NH3��+2H2O��
�ʴ�Ϊ��2NH4++SO42-+Ba2++2OH-
BaSO4��+2NH3��+2H2O��
�ʵ�λ��SO42-��SO32-��CO32-��
�ۼ���Ũ����������Һ�����ȣ������Թܿ���ʪ��ĺ�ɫʯ����ֽ���飬��ֽ������֤����������ӣ�
�ʴ�Ϊ��NH4+��
�������Ϸ�������ɫ�������Ϊ������NH4��2SO4��NH4��2SO3��NH4��2CO3
�ʴ�Ϊ����NH4��2SO4��NH4��2SO3��NH4��2CO3��
��2�����һ��ȷ�ϴ˰�ɫ����ijɷ֣���Ҫ�ڲ���ڼ���������м��飬
�ʴ�Ϊ���ڣ����
��3�����ڼ���������Ȼ���ɳ���������Ϊ���ᱵ����ɫ����Ϊ����泥���Ӧ�����ӷ���ʽΪ��2NH4++SO42-+Ba2++2OH-
| ||
�ʴ�Ϊ��2NH4++SO42-+Ba2++2OH-
| ||
���������⿼����δ֪��ļ��飬���������ƶ��⣬ע�����������Ϣ�����ƶϣ������Ѷ��еȣ�

��ϰ��ϵ�д�
 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�
�����Ŀ