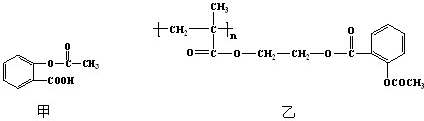
��Ŀ����
��1��SiO2��
��2��������ѧʽ
��3���嵥�ʣ������ӣ��ⵥ�ʣ�������ͬʱ�ӵ�ͬһ�ձ��е����ӷ���ʽ
��4���α��ϳ��ֵĿ��淴Ӧһ���Ƕ�����������ˮ������������ҵӦ�ù㷺�ķ���ʽΪ
��״
��״
�ṹ����������������������ѧ���ʣ���2��������ѧʽ
KAl��SO4��2?12H2O
KAl��SO4��2?12H2O
��3���嵥�ʣ������ӣ��ⵥ�ʣ�������ͬʱ�ӵ�ͬһ�ձ��е����ӷ���ʽ
Br2+2I-=2Br-+I2
Br2+2I-=2Br-+I2
��4���α��ϳ��ֵĿ��淴Ӧһ���Ƕ�����������ˮ������������ҵӦ�ù㷺�ķ���ʽΪ
N2+3H2
2NH3��2SO2+O2
2SO3
| ||
| ���¸�ѹ |
| ||
| �� |
N2+3H2
2NH3��2SO2+O2
2SO3
��
| ||
| ���¸�ѹ |
| ||
| �� |
��������1��SiO2������״�ṹ��
��2��������ʮ��ˮ���������أ�
��3���嵥���ܽ�����������Ϊ�ⵥ�ʣ�
��4���ϳɰ�����ȡ���������ǿ��淴Ӧ��
��2��������ʮ��ˮ���������أ�
��3���嵥���ܽ�����������Ϊ�ⵥ�ʣ�
��4���ϳɰ�����ȡ���������ǿ��淴Ӧ��
����⣺��1��SiO2����״�ṹ�����������������������ͻ�ѧ���ʣ��ʴ�Ϊ����״��
��2��������ʮ��ˮ���������أ���ѧʽ��KAl��SO4��2?12H2O���ʴ�Ϊ��KAl��SO4��2?12H2O��
��3���嵥���ܽ�����������Ϊ�ⵥ�����ӷ���ʽ��Br2+2I-=2Br-+I2���ʴ�Ϊ��Br2+2I-=2Br-+I2��
��4��������ҵӦ�ù㷺�ķ���ʽΪN2+3H2
2NH3��2SO2+O2
2SO3���ʴ�Ϊ��N2+3H2
2NH3��2SO2+O2
2SO3��
��2��������ʮ��ˮ���������أ���ѧʽ��KAl��SO4��2?12H2O���ʴ�Ϊ��KAl��SO4��2?12H2O��
��3���嵥���ܽ�����������Ϊ�ⵥ�����ӷ���ʽ��Br2+2I-=2Br-+I2���ʴ�Ϊ��Br2+2I-=2Br-+I2��
��4��������ҵӦ�ù㷺�ķ���ʽΪN2+3H2
| ||
| ���¸�ѹ |
| ||
| �� |
| ||
| ���¸�ѹ |
| ||
| �� |
������������Ҫ�������ʵ����ʡ�����Լ���;����Ŀ�ѶȲ���ѧϰ��ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
����������ȷ���ǣ�������
| A��SiO2������״�ṹ�У���С�Ļ��ϣ�Si��Oԭ����֮��Ϊ1��2 | B����������Ԫ�ص���������ϼ۵������������������� | C��SO2��SO3���Ǽ��Է��� | D��ͬ����Ԫ���γɵ�������ľ������;���ͬ |