��Ŀ����
�ȼ��ⱥ��ʳ��ˮ��ȡNaOH��Һ�Ĺ���������ͼ��ʾ������������

(1)�ڵ������У����Դ���������ĵ缫�ϵĵ缫��ӦʽΪ_______________�����������Դ���������ĵ缫�ϵ�������ʵ������ͨ��ѡ�õĻ�ѧ�Լ���________��
(2)��ҵʳ���к�Ca2+��Mg2+�����ʣ����ƹ��̷�����Ӧ�����ӷ���ʽΪ________________��
(3)���������SO42-�����ϸߣ��������ӱ��Լ���ȥ���ñ��Լ�������________������ĸ��ţ���
a. Ba(OH)2 b.Ba(NO3)2 c.BaCl2
(4)Ϊ����Ч�س�ȥCa2+��Mg2+��SO42-�������Լ��ĺ���˳��Ϊ_______������ĸ��ţ���
a���ȼ�NaOH�����Na2CO3���ټӱ��Լ�
b���ȼ�NaOH����ӱ��Լ����ټ�Na2CO3
c���ȼӱ��Լ������NaOH���ټ�Na2CO3
(4)���ι���������NaOH��NaCl���ܽ���ϵIJ��죬ͨ��______����ȴ��______����д�������ƣ���ȥ
NaCl��
(2)��ҵʳ���к�Ca2+��Mg2+�����ʣ����ƹ��̷�����Ӧ�����ӷ���ʽΪ________________��
(3)���������SO42-�����ϸߣ��������ӱ��Լ���ȥ���ñ��Լ�������________������ĸ��ţ���
a. Ba(OH)2 b.Ba(NO3)2 c.BaCl2
(4)Ϊ����Ч�س�ȥCa2+��Mg2+��SO42-�������Լ��ĺ���˳��Ϊ_______������ĸ��ţ���
a���ȼ�NaOH�����Na2CO3���ټӱ��Լ�
b���ȼ�NaOH����ӱ��Լ����ټ�Na2CO3
c���ȼӱ��Լ������NaOH���ټ�Na2CO3
(4)���ι���������NaOH��NaCl���ܽ���ϵIJ��죬ͨ��______����ȴ��______����д�������ƣ���ȥ
NaCl��
(1)2H+ +2e-=H2������2H2O +2e-=H2��+2OH-)�����ۡ��⻯�أ�����ۡ��⻯����Һ��
(2)Ca2++CO32-=CaCO3����Mg2++2OH-=Mg(OH)2��
(3)ac
(4)bc
(5)��������������
(2)Ca2++CO32-=CaCO3����Mg2++2OH-=Mg(OH)2��
(3)ac
(4)bc
(5)��������������

��ϰ��ϵ�д�
�����Ŀ
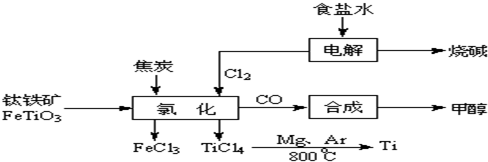
 2OH-+H2��+Cl2��
2OH-+H2��+Cl2�� 2MgCl2��s��+Ti����Ar�����н��е������ǣ�
2MgCl2��s��+Ti����Ar�����н��е������ǣ�





 ____________
____________

 ��Ar������������________
��Ar������������________