��Ŀ����
��15�֣�ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϡ�
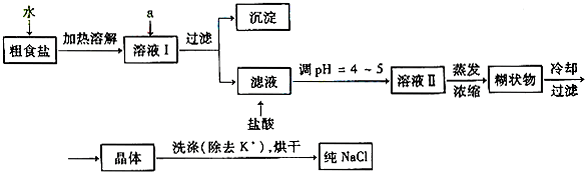
(1)��ʳ�γ���������K+��Ca2+��Mg2+��Fe3+��SO42�����������ӣ�ʵ�����ᴿNaCl���������£�
�ṩ���Լ�������Na2CO3��Һ ����K2CO3��Һ NaOH��Һ BaCl2��Һ Ba(NO3)2��Һ 75%�Ҵ������Ȼ�̼
������ȥ��ҺI�е�Ca2+��Mg2+��Fe3+��SO42�����ӣ�ѡ��a���������Լ������μ�˳������Ϊ_______ __��ֻ�ѧʽ����
��ϴ�ӳ�ȥNaCl������渽��������KCl��ѡ�õ��Լ�Ϊ_____ ____��
(2)���ᴿ��NaCl����500 mL4.00mol��L-1NaCl��Һ��������������ƽ��ҩ�ס����������___ _____�����������ƣ���
(3) ��ⱥ��ʳ��ˮ��װ����ͼ��ʾ��
���ռ���H2Ϊ2 L��
��ͬ���������ռ���Cl2______���>������=����<����2 L��ԭ����__________��װ�øĽ��������Ʊ�NaOH��Һ�����ⶨ��Һ��NaOH��Ũ�ȣ����õķ�����_____________��
(4)ʵ�����Ʊ�H2��Cl2ͨ���������з�Ӧ��Zn+H2SO4 ZnSO4+H2����MnO2+4HCl��Ũ��MnCl2+Cl2��+2H2O���ݴˣ���������������װ����ѡ���Ʊ����ռ�H2��װ��_______(�����)���Ʊ����ռ��������Cl2��װ��_________������ţ���
��ѡ���Ʊ������װ�ã�
����:��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�