��Ŀ����
ͨ��ʵ�顢�۲졢��ȡ������ȷ����ó���ȷ�Ľ����ǻ�ѧѧϰ�ķ���֮һ�������з�Ӧ���ƶϻ������ȷ����(����)
| | ���� | ���ܵ�ʵ������ | ���� |
| A | ij��Һ�м��������ữ���Ȼ�����Һ | �а�ɫ�������� | ��Һ��һ������SO |
| B | ��CaSO3�м������� | �д̼�����ζ���������� | CaSO3�����ᷴӦ����SO2 |
| C | ͬ��ͬѹ����pH��ֽ�ⶨ��ͬ Ũ�ȵ�̼���ƺ���������Һ������� Ũ�ȵ�̼���ƺ���������Һ������� | ̼������Һ�Լ��ԣ���������Һ������ | ̼������Һ������ˮ�� |
| D | �����KI��Һ��ͨ��Cl2 | ��Һ���� | Cl2����۷�����ɫ��Ӧ |
C
��

��ϰ��ϵ�д�
 ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�
�����Ŀ
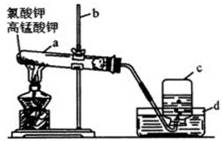

 ����������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ���ѡ�Լ���3mol��L��1H2SO4��6 mol��L��1NaOH��0.5 mol��L��1BaCl2��0.5 mol��
����������̽����ʵ���������ɷ֡������ʵ����ƣ���д�����Լ���Ԥ����������ۡ���ѡ�Լ���3mol��L��1H2SO4��6 mol��L��1NaOH��0.5 mol��L��1BaCl2��0.5 mol�� L��1Ba(NO3)2��0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ
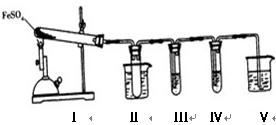
L��1Ba(NO3)2��0.01 mol��L��1����KMnO4��Һ��0.01 mol��L��1��ˮ CO�� + CO2�� + H2O��
CO�� + CO2�� + H2O��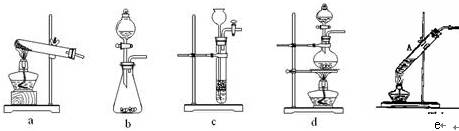
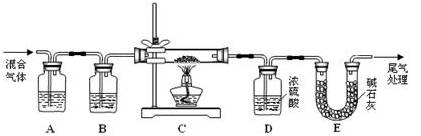
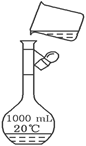
 ��������ȷ��������ʱ�����ӿ̶��ߣ���������ҺŨ��_____0.8 mol/L���������������ͬ����������ʱ������������ˮ����������ƿ�⣬��������ҺŨ��_____0.8 mol/L��
��������ȷ��������ʱ�����ӿ̶��ߣ���������ҺŨ��_____0.8 mol/L���������������ͬ����������ʱ������������ˮ����������ƿ�⣬��������ҺŨ��_____0.8 mol/L�� ���Ȳ���������Ȼ��������ܽ⡱������ǣ� ��
���Ȳ���������Ȼ��������ܽ⡱������ǣ� �� �IJ��������ҳ�����ͼʾ����ȷ��ʵ����� �� ��
�IJ��������ҳ�����ͼʾ����ȷ��ʵ����� �� ��