��Ŀ����
��14�֣���Ӧ�� Fe(s)+CO2(g)===FeO(s)+CO(g)����H1=" a" kJmol-1��Ӧ�� CO(g)+1/2O2(g)===CO2(g)����H2=" b" kJmol-1����ڲ�ͬ�¶��£����ܱ������н��еķ�Ӧ�ٵ�ƽ�ⳣ��Kֵ���¶ȵı仯���£�
(1)��Ӧ�ٵĻ�ѧƽ�ⳣ������ʽΪ ��a 0���>������<����=�������� 500��2L �ܱ������н��з�Ӧ�٣�Fe �� CO2����ʼ���ʵ�����Ϊ4mol��10min��ﵽƽ��״̬��ƽ��ʱCO2��ת����Ϊ ����CO��ʾ�Ļ�ѧ��Ӧ����Ϊ ��
(2)700�淴Ӧ�ٵ�ƽ�⣬Ҫʹ��ƽ�������ƶ���������������ʱ�����Բ�ȡ�Ĵ�ʩ��__________������ţ���
| A����С��Ӧ������� | B��ͨ��CO2 |
| C�������¶ȵ�900�� | D��ʹ�ú��ʵĴ��� |
(4)��ͼ�Ǽ״�ȼ�ϵ�أ��������ҺΪKOH��Һ���Ľṹʾ��ͼ����a��ͨ����� ����״���������������b���缫�Ϸ����ĵ缫��Ӧʽ��

��1��K=c(CO)/c(CO2)��1�֣� >��1�֣� 50%��1�֣� 0.1mol/(L��min��1) ��1�֣�
��2��BC��2�֣� ��3��2��a+b��kJ��mol-1��3�֣�
��4��������2�֣� CH3OH �� 6e- + 8OH - ="=" CO3 2- + 6H2O (3��)
����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
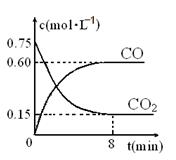
Сѧ��10����Ӧ����ϵ�д�һ�������������Ժ�CO2������Ӧ��Fe(s)��CO2(g) FeO(s)��CO(g)��H��0��1100��ʱ����ij�ܱ������м����������۲�����һ������CO2���壬��Ӧ������CO2�����CO�����Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��
FeO(s)��CO(g)��H��0��1100��ʱ����ij�ܱ������м����������۲�����һ������CO2���壬��Ӧ������CO2�����CO�����Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��
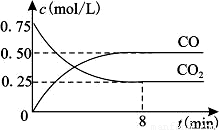
��1�����д�ʩ����ʹƽ��ʱK�������___________������ţ���
A�������¶�??? B������ѹǿC������һ����COD�������¶�
��2��8�����ڣ�CO��ƽ����Ӧ����v��CO��=___________mol/(L��min)��
��3��1100��ʱ��2L���ܱ������У�����ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���÷�Ӧ�ﵽƽ��ʱ���й���������
���� | �� | �� |
��Ӧ��Ͷ���� | 3molFe��2molCO2 | 4molFeO��3molCO |
CO��Ũ��(mol/L) | C1 | C2 |
CO2��������� |
|
|
��ϵѹǿ��Pa�� | P1 | P2 |
��̬��Ӧ���ת���� |
|
|
������˵����ȷ����___________��
A��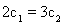 B��
B��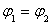 C��P1��P2D��
C��P1��P2D��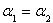
����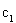 ��___________��
��___________��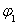 ��___________��
��___________��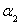 ��___________��
��___________��
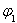
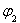
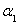
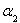
 FeO(s)+CO(g)����H>0
FeO(s)+CO(g)����H>0
 FeO(s)+CO(g)����H>0
FeO(s)+CO(g)����H>0