��Ŀ����
| |||||||||||||||||||
������
(1) |
ÿ��һ����Ŀ��Ԫ�أ�����Ԫ�صı仯�ظ�ǰ��Ԫ�ر仯�Ĺ��� |
(2) |
2.0���֣�2.4 |
(3) |
�������ӵ����� |
(4) |
|

H:0.1 |
|
|
|
|
|
|
Li:1.0 | Be:1.5 | B:2.0 | C:2.5 | N:3.0 | O:3.5 | F:4.0 |
Na:0.9 | Mg:1.2 | Al:1.5 | Si:1.8 | P:2.1 | S:2.5 | Cl:3.0 |
K:0.8 | Ca:1.0 | Ga:1.6 | Ge:1.8 | As:2.0 | Se:2.4 | Br:2.8 |
Rb:0.8 | Sr:1.0 | In:1.7 | Sn:1.8 | Sb:1.9 | Te:�� | I:2.5 |
Cs:0.7 | Ba:0.9 | Tl:1.8 | Pb:1.9 | Bi:1.9 | Po:2.0 | At:2.5 |
Fr:0.7 | Ra:0.9 |
|
|
|
|
|
�ش��������⣺
(1)д������Ԫ�ص縺����ͬ���ڻ�ͬ�����еĵݱ����(�ζ�һ��)��___________________��
(2)Ԥ��TeԪ�ئֵ�ֵΪ_________�����ķǽ����Ա�IԪ��_________ (ѡ�ǿ��������)��
(3)����ΪLinus Pauling����縺�Եĸ�����ȷ����������ԭ��_________(ѡ�ʧ���ӡ��õ��ӡ�)��������Դ�С��
(4)������ʵ������������Ԫ�صĦ�ֵ�����ڻ����1.7ʱ���γɵĻ�����һ�������ӻ�
������ݴ˾�����ɣ�AlCl3�л�ѧ��������Ӧ����_________��
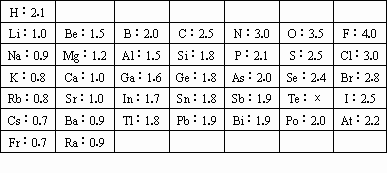
1932�꣬������ѧ��ʦLinus Pauling����縺�ԣ���ϣ����ĸ�ֱ�ʾ���ĸ������ȷ����������ԭ��ij����������Դ�С��Linus Pauling�ٶ�F�ĵ縺��Ϊ4����ͨ���Ȼ�ѧ��������������Ԫ�صĵ縺�ԡ�Linus Pauling����������Ԫ�صĵ縺�����£�
| H��2.1 | | | | | | |
| Li��1.0 | Be��1.5 | B��2.0 | C��2.5 | N��3.0 | O��3.5 | F��4.0 |
| Na��0.9 | Mg��1.2 | Al��1.5 | Si��1.8 | P��2.1 | S��2.5 | Cl��3.0 |
| K��0.8 | Ca��1.0 | Ga��1.6 | Ge��1.8 | As��2.0 | Se��2.4 | Br��2.8 |
| Rb��0.8 | Sr��1.0 | In��1.7 | Sn��1.8 | Sb��1.9 | Te���� | I��2.5 |
| Cs��0.7 | Ba��0.9 | Tl��1.8 | Pb��1.9 | Bi��1.9 | Po��2.0 | At��2.2 |
| Fr��0.7 | Ra��0.9 | | | | | |
�ش��������⣺
���ݹ۸���������Ԫ�ص縺�Ա仯��̸̸���Ԫ�����ʳ��������Ա仯�����⣺ ��
��Ԥ��TeԪ�ئֵ�ֵ ��
������ΪLinus Pauling����縺�Եĸ�����ȷ����������ԭ��������������Դ�С�� ��
�ȴ�����ʵ������������Ԫ�صĵ縺�Բ�ֵС��1.7ʱ��������Ԫ��ͨ���γɹ��ۻ�����õ���ʽ��ʾAlBr3���γɹ��� ��
��8�֣��ơ�þ��������Ҫ�Ľ���Ԫ�أ������ǵ������������ϵ���С�
��1��Ԫ�ص������ر��ǻ�ѧ����ȡ����Ԫ��ԭ�ӽṹ�������ӵĵ����Ų�ʽΪ_______����Ԫ�ص�ԭ�ӽṹʾ��ͼΪ_________��
��2���ơ�þ����Ԫ�ص������Ӱ뾶��С�����˳����_____________(�����ӷ��ű�ʾ)���ڶ������зǽ�������ǿ��Ԫ��λ�ڵ�____����____�塣
��3���ơ�þ�����ĵ��ʼ��仯������ijЩ�����ϴ����ŵݱ���ɡ������й�˵����ȷ����___________��ѡ���ţ���
a������Ԫ�صĽ�����Խǿ���������ʵ��۵��Խ��
b�������NaOH��Mg(OH)2��Al(OH)3˳�����μ���
c�������¶�����Ũ������ҷ�Ӧ���������Σ������ҳ̶����μ���
d�����������ơ�þ����������ϡ���ᷴӦ�������������ʵ�����������
��4��1932�꣬������ѧ��ʦLinus Pauling����縺�ԣ���ϣ����ĸ�ֱ�ʾ���ĸ������ȷ����������ԭ��ij����������Դ�С��Linus Pauling�ٶ���Ԫ�صĵ縺��Ϊ4����ͨ���Ȼ�ѧ��������������Ԫ�صĵ縺�ԡ�������������Ԫ�صĵ縺�����£�
|
Ԫ�� |
Na |
Mg |
Al |
Si |
P |
S |
Cl |
|
�縺�� |
0.9 |
1.2 |
1.5 |
1.8 |
2.1 |
2.5 |
3.0 |
���ϱ����Կ����縺�ԵĴ�С��Ԫ�طǽ����Ե�ǿ����ϵ��____________________��������ʵ������������Ԫ�صĦ�ֵ�����ڻ����1.7ʱ���γɵĻ�����һ�������ӻ�������ݴ˾�����ɣ�AlBr3�еĻ�ѧ������Ӧ���� ��
1932�꣬������ѧ��ʦLinus Pauling����縺�ԣ���ϣ����ĸ�ֱ�ʾ���ĸ������ȷ����������ԭ��ij����������Դ�С��Linus Pauling�ٶ�F�ĵ縺��Ϊ4����ͨ���Ȼ�ѧ��������������Ԫ�صĵ縺�ԡ�Linus Pauling����������Ԫ�صĵ縺�����£�
|
H��2.1 |
|
|
|
|
|
|
|
Li��1.0 |
Be��1.5 |
B��2.0 |
C��2.5 |
N��3.0 |
O��3.5 |
F��4.0 |
|
Na��0.9 |
Mg��1.2 |
Al��1.5 |
Si��1.8 |
P��2.1 |
S��2.5 |
Cl��3.0 |
|
K��0.8 |
Ca��1.0 |
Ga��1.6 |
Ge��1.8 |
As��2.0 |
Se��2.4 |
Br��2.8 |
|
Rb��0.8 |
Sr��1.0 |
In��1.7 |
Sn��1.8 |
Sb��1.9 |
Te���� |
I��2.5 |
|
Cs��0.7 |
Ba��0.9 |
Tl��1.8 |
Pb��1.9 |
Bi��1.9 |
Po��2.0 |
At��2.2 |
|
Fr��0.7 |
Ra��0.9 |
|
|
|
|
|
�ش��������⣺
���ݹ۸���������Ԫ�ص縺�Ա仯��̸̸���Ԫ�����ʳ��������Ա仯�����⣺ ��
��Ԥ��TeԪ�ئֵ�ֵ ��
������ΪLinus Pauling����縺�Եĸ�����ȷ����������ԭ��������������Դ�С�� ��
�ȴ�����ʵ������������Ԫ�صĵ縺�Բ�ֵС��1.7ʱ��������Ԫ��ͨ���γɹ��ۻ�����õ���ʽ��ʾAlBr3���γɹ��� ��