��Ŀ����
��8��)���ᡢ�������������ѧ�γ����ġ������ᡱ���־��������������Ӧ��������ش��������⣺
(1)ϡ�����Cu��Ӧ������ϡ�����м���H2O2����������������������ʱ��ԭ����Ϊˮ�������ʹͭ˳���ܽ⣬���õ���ɫ��Һ���÷�Ӧ�Ļ�ѧ����ʽΪ��_________________��
(2)��һ�������18 mol��L-1��Ũ�����м������ͭƬ������ʹ֮��Ӧ��������ԭ������Ϊ0.9mol����Ũ�����ʵ�����_________(����ڡ��������ڡ���С�ڡ�)100mL����ʹʣ���ͭƬ�����ܽ⣬�������м�����������Һ����KNO3��Һ������÷�Ӧ�����ӷ���ʽΪ__________________________________��
��3��������ͼ�����������ƶ���XΪ______________(�����)��
a��Ũ���� b��Ũ���� c��Ũ����
��ÿ��2�֡���8�֣�
��1��Cu+H2O2+2HCl��CuCl2+2H2O
��2�����ڣ� 3Cu+8H++2NO3-��3Cu2++2NO +4H2O
����

��ϰ��ϵ�д�
�����Ŀ

 CuO
CuO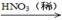 Cu(NO3)2
Cu(NO3)2

