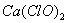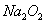��Ŀ����
��13�֣���84������Һ��������������֪ijƷ�ơ�84������Һ����Ч�ɷ�ΪNaClO������нϴ�����Na2CO3��NaCl��Ϊ̽��������Һ�����ʣ�ijͬѧ��������ʵ�飺ȡ��������Һ��������ͨ�������CO2���ٵ���Ʒ�죬����Ʒ��ܿ���ɫ��
��1��д������Һ��ͨ�������CO2���������������ӷ���ʽ��
��������������������������������������������������������������������������
������������������������������������������������������������������������2��ʹƷ��ܿ���ɫ��������������������������
��3���Ը�����ҺΪ��Ҫ��Դ�����Ʊ��ϴ���������Cl2�����������������Ҫ���裺��������Һ�м��������A�Լ������ú�ȡ�ϲ���Һ���ټ��������ᡣ �����ѣ�������ķ�ӦΪ��NaCl + NaClO + H2SO4 = Cl2��+ Na2SO4 +H2O)
�� A�Լ��Ļ�ѧʽΪ_____________����������������������������������
�� �����ɱ������5.6 L Cl2����ת�Ƶ��ӵ����ʵ���Ϊ____________��
��1��д������Һ��ͨ�������CO2���������������ӷ���ʽ��
��������������������������������������������������������������������������
������������������������������������������������������������������������2��ʹƷ��ܿ���ɫ��������������������������
��3���Ը�����ҺΪ��Ҫ��Դ�����Ʊ��ϴ���������Cl2�����������������Ҫ���裺��������Һ�м��������A�Լ������ú�ȡ�ϲ���Һ���ټ��������ᡣ �����ѣ�������ķ�ӦΪ��NaCl + NaClO + H2SO4 = Cl2��+ Na2SO4 +H2O)
�� A�Լ��Ļ�ѧʽΪ_____________����������������������������������
�� �����ɱ������5.6 L Cl2����ת�Ƶ��ӵ����ʵ���Ϊ____________��
25����13�֣�
��1��ClO- + CO2 + H2O ��HClO + HCO3?��2�֣�
H2O + CO2 + CO32-��2 HCO3?��2�֣� HClO��2�֣�
��2�� ��CaCl2����BaCl2��2�֣��� ��ȥCO32-��2�֣� �� 0.25 mol��3�֣�
��1��ClO- + CO2 + H2O ��HClO + HCO3?��2�֣�
H2O + CO2 + CO32-��2 HCO3?��2�֣� HClO��2�֣�
��2�� ��CaCl2����BaCl2��2�֣��� ��ȥCO32-��2�֣� �� 0.25 mol��3�֣�
�����������84������Һ����Ч�ɷ�ΪNaClO������Һ��ͨ�������CO2ʱ��Ӧ�Ļ���ԭ����ǿ�������ᣬ���Է������������ӷ���ʽ��ClO- + CO2 + H2O ��HClO + HCO3? �����нϴ�����Na2CO3���÷�Ӧ��̼���ƺ�ת����̼�����ƵĹ��̣� H2O + CO2 + CO32-��2 HCO3?��ʹƷ��ܿ���ɫ��������HClO���Ը�����ҺΪ��Ҫ��Դ�����Ʊ��ϴ���������Cl2�����������������Ҫ���裺��������Һ�м��������CaCl2����BaCl2�Լ�����ȥCO32-�����ú�ȡ�ϲ���Һ���ټ�������������ɱ������5.6 L Cl2����ת�Ƶ��ӵ����ʵ���Ϊ0.25 mol������ļ�����������£�NaCl + NaClO + H2SO4 = Cl2��+ Na2SO4 +H2Oÿ����1mol�����ͻ�ת��1mol����������5.6L�����õ����������ʵ�����0.25mol����ת�Ƶĵ�����0.25mol��
������������ѧ��֪ʶ���ʵ��������ѧ������Ҫ�ﵽ��Ŀ�꣬���Դ�����Ŀ��Ҫ����ѧ������ѧ֪ʶ����������������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ