��Ŀ����
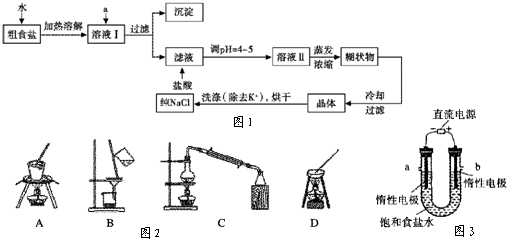
(10��)��ʳ�γ���������Ca2+��Mg2+��SO42-�Լ���ɳ�����ʣ��������ᴿNaCl���������£�������Լ��Թ�����
 |
(1)������м�����Լ�A�� (�ѧʽ)��
(2)������У���ص����ӷ���ʽ
��
(3)����������벽��Ե������������pH�ٹ��ˣ�����ʵ����������Ӱ����
��
(4)���ᴿ��NaC1����100mL 1.0mol��L��NaCl��Һ�������������ձ�������������ͷ�ιܡ�ҩ���⣬����Ҫ (����������)��
(5)ʵ�����ᴿNaCl�Ĺ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ�������������������ʱ��ʹ�ò�������Ŀ�� ��
(1)NaOH (2��)
(2)Ca2++CO32- ��CaCO3�� (1��)�� Ba2++CO32-��BaCO3��(1��)
[��ѧʽ�������֣�ûд����������1��]
(3)�ȼ�Hcl��������CaCO3��Ba(��03��Mg(OH)2�����ܽ⣬ʹNaCl�������ʣ��Ӷ�Ӱ���Ƶ�NaCl�Ĵ��ȡ� (2��)
(4)100mL����ƿ(1��)����ƽ(1��)
(5)��ֹ�ֲ�����Һ�λ���ɽ��� (2��) ![]()

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�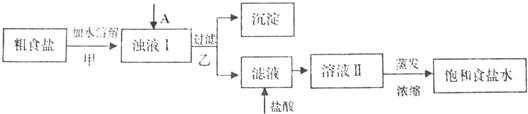

 2 L��ԭ���� ��
2 L��ԭ���� ��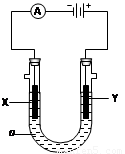
 2
L��ԭ����
��
2
L��ԭ����
��