��Ŀ����
A��B��C��D��E���Ƕ���������Ԫ�أ�ԭ��������������B��Cͬ���ڣ�A��Dͬ���壮A��B���γ�����Һ̬��������ң�ԭ�Ӹ����ȷֱ�Ϊ2��1��1��1������������Ϣ�ش��������⣺
��1���ס����������к��зǼ��Թ��ۼ���������______���ѧʽ����CԪ�������ڱ��е�λ����______��
��2��C��D�������У��뾶��С����______�������ӷ��ţ���
��3���õ���ʽ��ʾD2B ���γɹ���______
��4����D�ĵ���Ͷ����У���D��ʧ������������Һ�м��뵥��E��������A2��������E������______����дԪ�ط��ţ������ɸ���������ӷ���ʽ��______��
��1���ס����������к��зǼ��Թ��ۼ���������______���ѧʽ����CԪ�������ڱ��е�λ����______��
��2��C��D�������У��뾶��С����______�������ӷ��ţ���
��3���õ���ʽ��ʾD2B ���γɹ���______
��4����D�ĵ���Ͷ����У���D��ʧ������������Һ�м��뵥��E��������A2��������E������______����дԪ�ط��ţ������ɸ���������ӷ���ʽ��______��
A��B��C��D��E���Ƕ���������Ԫ�أ�ԭ��������������A��B���γ�����Һ̬��������ң�ԭ�Ӹ����ȷֱ�Ϊ2��1��1��1����AΪH��BΪO��A��Dͬ���壬��DΪNa��B��Cͬ���ڣ���CΪF����ϣ�4��Al��Ӧ������������EΪAl��
��1����ΪH2O��Ϊ�����Լ��ļ��Է��ӣ���ΪH2O2��Ϊ�����Լ����Ǽ��Լ��ļ��Է��ӣ�CΪF��λ�ڵڶ�����VIIA���ʴ�Ϊ��H2O2���ڶ�����VIIA��
��2��D2BΪNa2O���õ���ʽ��ʾ���γɹ���Ϊ

��
�ʴ�Ϊ��

��
��3��Na��ˮ��Ӧ����NaOH��E��NaOH��Һ��Ӧ������������EΪAl�����ӷ�ӦΪ2Al+2OH-+2H2O=2AlO2-+3H2�����ʴ�Ϊ��Al��2Al+2OH-+2H2O=2AlO2-+3H2����
��1����ΪH2O��Ϊ�����Լ��ļ��Է��ӣ���ΪH2O2��Ϊ�����Լ����Ǽ��Լ��ļ��Է��ӣ�CΪF��λ�ڵڶ�����VIIA���ʴ�Ϊ��H2O2���ڶ�����VIIA��
��2��D2BΪNa2O���õ���ʽ��ʾ���γɹ���Ϊ

��
�ʴ�Ϊ��

��
��3��Na��ˮ��Ӧ����NaOH��E��NaOH��Һ��Ӧ������������EΪAl�����ӷ�ӦΪ2Al+2OH-+2H2O=2AlO2-+3H2�����ʴ�Ϊ��Al��2Al+2OH-+2H2O=2AlO2-+3H2����

��ϰ��ϵ�д�
�����Ŀ
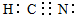
 CH3COOH+OH-
CH3COOH+OH- ��֪A��B��C��D��E����Ԫ�����ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��Bԭ�ӵ�p�����������γɵ��⻯��ķе���ͬ����Ԫ�ص��⻯������͵ģ�Dԭ�ӵõ�һ�����Ӻ�3p���ȫ������A+��Dԭ���γɵ�������һ�����Ӳ㣮C��A�γ�A2C�����ӻ����E��ԭ������Ϊ26��Eԭ�ӻ�������Χ�н϶���������Ŀչ��������һЩ���ӻ������γ��������������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
��֪A��B��C��D��E����Ԫ�����ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��Bԭ�ӵ�p�����������γɵ��⻯��ķе���ͬ����Ԫ�ص��⻯������͵ģ�Dԭ�ӵõ�һ�����Ӻ�3p���ȫ������A+��Dԭ���γɵ�������һ�����Ӳ㣮C��A�γ�A2C�����ӻ����E��ԭ������Ϊ26��Eԭ�ӻ�������Χ�н϶���������Ŀչ��������һЩ���ӻ������γ��������������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ�� ʾ��λ�ڸ�������Ķ�������ģ��û�����Ļ�ѧʽ��
ʾ��λ�ڸ�������Ķ�������ģ��û�����Ļ�ѧʽ��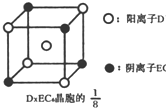
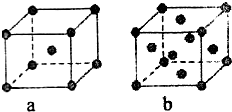 ��֪A��B��C��D��E����Ԫ�����ڱ��е�ǰ������Ԫ�أ�����ԭ�������Ĵ�С��ϵΪA��C��B��D��E����֪Aԭ�ӵ�p���Ϊ����������γɵ��⻯��ķе���ͬ����ǽ���Ԫ�ص��⻯������ߵģ�Dԭ�ӵõ�һ�����Ӻ���3p�����ȫ������B+���ӱ�Dԭ���γɵ�������һ�����Ӳ㣮C��B���γ�BC�͵����ӻ����E��ԭ������Ϊ29��
��֪A��B��C��D��E����Ԫ�����ڱ��е�ǰ������Ԫ�أ�����ԭ�������Ĵ�С��ϵΪA��C��B��D��E����֪Aԭ�ӵ�p���Ϊ����������γɵ��⻯��ķе���ͬ����ǽ���Ԫ�ص��⻯������ߵģ�Dԭ�ӵõ�һ�����Ӻ���3p�����ȫ������B+���ӱ�Dԭ���γɵ�������һ�����Ӳ㣮C��B���γ�BC�͵����ӻ����E��ԭ������Ϊ29��