��Ŀ����
��25��ʱ����ʯī�缫���1.0L 2.5mol/LCuSO4��Һ��5min����һ��ʯī�缫����3.2g Cu���ɣ��Իش��������⣺
��1����д�����CuSO4��Һ�Ļ�ѧ����ʽ______��
��2����______mol���ӷ���ת�ƣ��õ�O2���������״������______mL����Һ��PH��______��
��3�����õ�����������ͭƬ����ʯī���缫�������ͬʱ�����ͭƬ���������______g�����Һ��pH______�����С����������䡱��
��1����д�����CuSO4��Һ�Ļ�ѧ����ʽ______��
��2����______mol���ӷ���ת�ƣ��õ�O2���������״������______mL����Һ��PH��______��
��3�����õ�����������ͭƬ����ʯī���缫�������ͬʱ�����ͭƬ���������______g�����Һ��pH______�����С����������䡱��
��1���������ͭʱ�����������������ŵ磬��������ͭ���ӷŵ磬
���ԭ������ʽΪ��2CuSO4+2H2O=2Cu+O2��+2H2SO4���ʴ�Ϊ��2CuSO4+2H2O=2Cu+O2��+2H2SO4��
��2���������ͭʱ����������ͭ���ӷŵ磬��Cu2++2e-��Cu�����õ缫����3.2g Cu����ʱ��ת�Ƶ���Ϊ
��2=0.1mol�����������������ŵ磬��4OH-��O2��+2H2O+4e-����ת��0.1mol����ʱ�����������������Ϊ��
��22.4L/mol=0.56L=560mL�����ٵ���������Ũ��Ϊ��
=0.1mol/L�����Ե���������Ũ��Ϊ0.1mol/L����Һ��pHΪ1���ʴ�Ϊ��0.1��560��1��
��3�����õ�����������ͭƬ����ʯī���缫��������ӦʽΪ��Cu��Cu2++2e-����ת��0.1mol����ʱ���ü���������3.2g��������ӦʽΪ��Cu2++2e-��Cu����ת��0.1mol����ʱ���ü���������3.2g��������ͭƬ���������6.4g�����Һ��pH���ֲ��䣮�ʴ�Ϊ��6.4�����䣮
���ԭ������ʽΪ��2CuSO4+2H2O=2Cu+O2��+2H2SO4���ʴ�Ϊ��2CuSO4+2H2O=2Cu+O2��+2H2SO4��
��2���������ͭʱ����������ͭ���ӷŵ磬��Cu2++2e-��Cu�����õ缫����3.2g Cu����ʱ��ת�Ƶ���Ϊ
| 3.2g |
| 64g/mol |
| 0.1mol |
| 4 |
| 0.1mol |
| 1.0L |
��3�����õ�����������ͭƬ����ʯī���缫��������ӦʽΪ��Cu��Cu2++2e-����ת��0.1mol����ʱ���ü���������3.2g��������ӦʽΪ��Cu2++2e-��Cu����ת��0.1mol����ʱ���ü���������3.2g��������ͭƬ���������6.4g�����Һ��pH���ֲ��䣮�ʴ�Ϊ��6.4�����䣮

��ϰ��ϵ�д�
 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
�����Ŀ
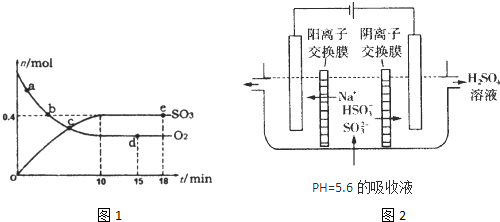
 Pa�£��ݻ�Ϊ1��0L���ܱ������г���0.5molSO2,
(g)��0.3 molO2 (g)��������Ӧ2SO2(g)��O2(g)
Pa�£��ݻ�Ϊ1��0L���ܱ������г���0.5molSO2,
(g)��0.3 molO2 (g)��������Ӧ2SO2(g)��O2(g) 2SO3(g)
��H����198kJ/mol����Ӧ��n(SO3)��n(O2)��ʱ��仯�Ĺ�ϵ����ͼ��ʾ
2SO3(g)
��H����198kJ/mol����Ӧ��n(SO3)��n(O2)��ʱ��仯�Ĺ�ϵ����ͼ��ʾ ����Ӧ��ƽ�ⳣ��K��_______��0��10 min����SO2��ʾ��ƽ����Ӧ����_________������ͼ����Ϣ���ж�������������ȷ����_____������ţ���
����Ӧ��ƽ�ⳣ��K��_______��0��10 min����SO2��ʾ��ƽ����Ӧ����_________������ͼ����Ϣ���ж�������������ȷ����_____������ţ���