��Ŀ����
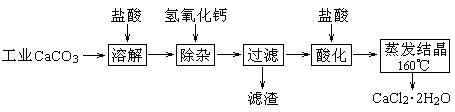
ҽ���Ȼ��ƿ������������ơ�������������ҩ��Թ�ҵ̼��ƣ����� ����Na+��Al3+��Fe3+�����ʣ�����ҽҩ����ˮ���Ȼ��ƣ�CaCl2��2H2O�������� ��Ϊ97.0%~103.0%������Ҫ�������£�

��1�����Ӳ����Ǽ����������ƣ�������Һ��pHΪ8.0һ8.5���Գ�ȥ��Һ�е����� Al3+��Fe3+������Fe(OH)3�Ƿ������ȫ��ʵ�������_____________________________________________��
��2���ữ�����Ǽ������ᣬ������Һ��pHԼΪ4.0����Ŀ���У��ٽ���Һ�е����� Ca( OH)2ת��ΪCaCl2���ڷ�ֹCa2+������ʱˮ�⣻��____��
��3���ⶨ��Ʒ��Cl-�����ķ����ǣ�a��ȡ0.7500 g��Ʒ���ܽ���250 mL����ƿ�ж��ݣ�b��ȡ25.00 mL������Һ����ƿ�У�c��0.05000 moI/LAgNO3��Һ�ζ����յ㣬����AgNO3��Һ�����ƽ��ֵΪ20. 39 mL��
�����ⶨ������������Һ��ϴ��������_______________ ��
�ڼ���������Ʒ��CaCl2��2H2O����������Ϊ___________________��
���������������ⶨ����Ʒ��CaCl2��2H2O����������ƫ�ߣ��ⶨ�����в��������ɺ��ԣ��������ԭ����___________________________��_____________________ ��
��2���ữ�����Ǽ������ᣬ������Һ��pHԼΪ4.0����Ŀ���У��ٽ���Һ�е����� Ca( OH)2ת��ΪCaCl2���ڷ�ֹCa2+������ʱˮ�⣻��____��
��3���ⶨ��Ʒ��Cl-�����ķ����ǣ�a��ȡ0.7500 g��Ʒ���ܽ���250 mL����ƿ�ж��ݣ�b��ȡ25.00 mL������Һ����ƿ�У�c��0.05000 moI/LAgNO3��Һ�ζ����յ㣬����AgNO3��Һ�����ƽ��ֵΪ20. 39 mL��
�����ⶨ������������Һ��ϴ��������_______________ ��
�ڼ���������Ʒ��CaCl2��2H2O����������Ϊ___________________��
���������������ⶨ����Ʒ��CaCl2��2H2O����������ƫ�ߣ��ⶨ�����в��������ɺ��ԣ��������ԭ����___________________________��_____________________ ��
��1��ȡ�����ϲ���Һ���μ�KSCN��Һ����������Ѫ��ɫ������Fe( OH)3������ȫ
��2����ֹ��Һ���տ����е�CO2
��3������ʽ�ζ��� ����99.9% ������Ʒ�д���������NaCl ������CaCl2��2H2Oʧˮ
��2����ֹ��Һ���տ����е�CO2
��3������ʽ�ζ��� ����99.9% ������Ʒ�д���������NaCl ������CaCl2��2H2Oʧˮ

��ϰ��ϵ�д�
 ͨ��ѧ��Ĭд����ϵ�д�
ͨ��ѧ��Ĭд����ϵ�д� ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�
�����Ŀ



 ���ⶨ��Ʒ��Cl-�����ķ����ǣ�a.��ȡ0.7500 g��Ʒ���ܽ⣬��250 mL����ƿ�ж��ݣ�b.��ȡ25.00 mL������Һ����ƿ�У�c.��0.05000 mol��L-1 AgNO3��Һ�ζ����յ㣬����AgNO3��Һ�����ƽ��ֵΪ20.39 mL��
���ⶨ��Ʒ��Cl-�����ķ����ǣ�a.��ȡ0.7500 g��Ʒ���ܽ⣬��250 mL����ƿ�ж��ݣ�b.��ȡ25.00 mL������Һ����ƿ�У�c.��0.05000 mol��L-1 AgNO3��Һ�ζ����յ㣬����AgNO3��Һ�����ƽ��ֵΪ20.39 mL��