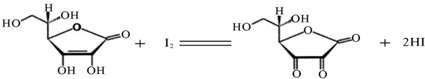��Ŀ����
��10�֣�ά����C��C6H8O6����Է�������Ϊ176��������ˮ������ǿ��ԭ�ԣ��ڼ�����Һ���ױ����������������Խ����н�Ϊ�ȶ���ά����Cͨ���ñ�I2��Һ��I2��ˮ���ܽ�Ⱥ�С��������KI��Һ��I2+I�C![]() I3�C���ʱ�I2��ҺӦ���I2��KI��Һ�����еζ���ԭ�����£�
I3�C���ʱ�I2��ҺӦ���I2��KI��Һ�����еζ���ԭ�����£�

��1���ζ�������Ӧѡ�� ������ʽ���ʽ���ζ���ʢ�ű�I2��Һ���ζ����õ�ָʾ��Ϊ ������ ����ʱ��Ϊ�ζ��յ㡣
��2������ά����C��Һ�����ƹ����У����Լ��������Լ��е� ������ĸ��
 A��0.1 mol��L-1 CH3COOH B��0.1 mol��L-1 HNO3
A��0.1 mol��L-1 CH3COOH B��0.1 mol��L-1 HNO3
C��0.1 mol��L-1 NaOH D��0.1 mol��L-1 Na2SO3
��3������һ�����ʵ���Ũ�ȵı�I2��Һ���貣�������� ������ĸ��
A������ƿ B����ͷ�ι� ��C�������� ��D���ձ� ������E����Ͳ
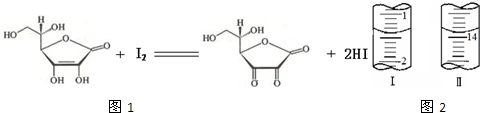
��4����1.0 g ά������Ʒ�Ƴ�100 mL��Һ��ȡ25 mL��0.1 mol��L-1��I2��Һ�ζ����ζ�ǰ��ζ�����ʾ����ͼ������Ʒ��ά����C����������Ϊ ��
(1)��ʽ��1�֣���������Һ��1�֣�����Һ����ɫ�պó��֣����ְ���Ӳ���ɫ��2�֣�����2��A��2�֣���3��ABCDE��ABCD��2�֣���4��88%��2�֣�

 ��У����ϵ�д�
��У����ϵ�д�| A������ | B�������� | C���л��� | D������ |
 I3-���ʱ�I2��ҺӦ���I2��KI��Һ�����еζ���ԭ�����£�
I3-���ʱ�I2��ҺӦ���I2��KI��Һ�����еζ���ԭ�����£�