��Ŀ����
��������һ�������մɲ��ϣ�������ʯӢ�뽹̿�ڸ��µĵ������У�ͨ�����·�Ӧ�Ƶã�
��SiO2����C����N2![]() ��Si3N4����CO
��Si3N4����CO
��1�� ��ƽ������Ӧ�Ļ�ѧ����ʽ������ѧ���������ڷ����ڣ���
��2���÷�Ӧ�е���������__________���仹ԭ������_____________��
��3���÷�Ӧ��ƽ�ⳣ������ʽΪK =_______________________��
��4����֪������ӦΪ���ȷ�Ӧ�����䷴Ӧ��DH________�㣨����ڡ�����С�ڡ����ڡ����������¶ȣ���ƽ�ⳣ��ֵ________�����������С�����䡱����
��5����ʹѹǿ����������ƽ����_______��Ӧ�����ƶ�����������桱����
��6������֪CO��������Ϊv ��CO�� =18 mol/��L?min������N2��������Ϊv ��N2�� =_________mol/��L?min����
��1��3��6��2��1��6 ��2��N2�� Si3N4 ��3��K =![]() ��4��С�ڣ���С
��4��С�ڣ���С
��5���� ��6��6

��ϰ��ϵ�д�
 ������״Ԫ��ҵϵ�д�
������״Ԫ��ҵϵ�д� ��ʱ�ƿ�������ϰϵ�д�
��ʱ�ƿ�������ϰϵ�д� һ��һ��һ��ͨϵ�д�
һ��һ��һ��ͨϵ�д�
�����Ŀ
 ��������һ�������մɲ��ϣ�������ʯӢ�����뽹̿�����ڸ��µĵ������У�ͨ�����·�Ӧ�Ƶã�3SiO2+6C+2N2
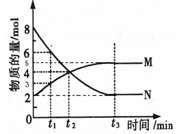
��������һ�������մɲ��ϣ�������ʯӢ�����뽹̿�����ڸ��µĵ������У�ͨ�����·�Ӧ�Ƶã�3SiO2+6C+2N2 Si3N4+6CO���÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⣺
Si3N4+6CO���÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⣺ ��������һ�������մɲ��ϵ���Ҫ�ɷ֣��ܳ��ܸ��£�����������ҵ������ҵ������ͼ�ǵ������ԭ�ӽṹʾ��ͼ�����Ʋ����Ļ�ѧʽ������ʽ��Ϊ��������
��������һ�������մɲ��ϵ���Ҫ�ɷ֣��ܳ��ܸ��£�����������ҵ������ҵ������ͼ�ǵ������ԭ�ӽṹʾ��ͼ�����Ʋ����Ļ�ѧʽ������ʽ��Ϊ�������� �÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⡣
�÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⡣ Si3N4 + 6CO���÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⣺
Si3N4 + 6CO���÷�Ӧ�����е������仯��ͼ��ʾ���ش��������⣺
 ������
������