��Ŀ����
��ҵ��ˮ�г�����һ������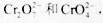 �����ǻ�����༰��̬ϵͳ�����ܴ���˺���������д��������õĴ������������֡�
�����ǻ�����༰��̬ϵͳ�����ܴ���˺���������д��������õĴ������������֡�
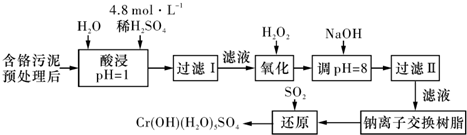
����1����ԭ������
�÷��Ĺ�������Ϊ

���еڢٲ�����ƽ�⣺

��1����ƽ����ϵ��pH=2������Һ�� ɫ.
��2����˵���ڢٲ���Ӧ��ƽ��״̬���� ��
a��Cr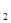 O
O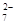
 ��CrO
��CrO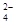 ��Ũ����ͬ
��Ũ����ͬ
b��2v (Cr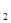 O
O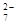 ) =v (CrO
) =v (CrO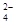 )
)
c.��Һ����ɫ����
��3���ڢڲ��У���ԭ1mol Cr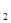 O
O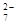 ���ӣ���Ҫ mol��FeSO
���ӣ���Ҫ mol��FeSO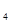 ��7H
��7H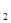 O��
O��
��4���ڢ۲����ɵ�Cr(OH)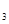 ����Һ�д������³����ܽ�ƽ�⣺
����Һ�д������³����ܽ�ƽ�⣺
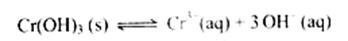
�����£�Cr(OH)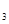 ���ܶȻ�
���ܶȻ� ��Ҫʹc��Cr3+������10
��Ҫʹc��Cr3+������10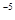 mol/L����Һ��pHӦ����
��
mol/L����Һ��pHӦ����
��
����2����ⷨ
�÷���Fe���缫��⺬Cr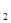 O
O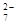 �����Է�ˮ�����ŵ����У�������������ҺpH���ߣ�����Cr(OH)
�����Է�ˮ�����ŵ����У�������������ҺpH���ߣ�����Cr(OH)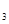 ������
������
��5����Fe���缫��ԭ��Ϊ ��
��6��������������ҺpH���ߵ�ԭ���ǣ��õ缫��Ӧ���ͣ� ��
��Һ��ͬʱ���ɵij������� ��

����������

��15�֣�ij��ҵ��ˮ�н����±������е�5�֣�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1mol/L��
|
������ |
K+ Cu2+ Fe3+ Al3+ Fe2+ |
|
������ |
Cl- CO32- NO3- SO42- SiO32- |
��ͬѧ��̽����ˮ����ɣ�����������ʵ�飺
���ò�˿պȡ������Һ���ڻ��������գ�����ɫ���棨����ɫ�ܲ����۲죩��
��ȡ������Һ������KSCN��Һ�����Ա仯��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬����Һ������������䡣
���� ��������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
��1���ɢ��жϣ���Һ��һ�������е��������� ��д���ӷ��ţ���
��2�����м�����������������ɫ����ĵ����ӷ���ʽ��_________________________��
��3�����������ú���ɫ����ͨ��ˮ�У��������ɫ���������Ļ�ѧ����ʽΪ
_______________________________________________________________
��4����ͬѧ����ȷ��ԭ��Һ�������������� ���������� ����д���ӷ��ţ�
��5����ȡ100mLԭ��Һ������������NaOH��Һ����ַ�Ӧ����ˣ�ϴ�ӣ����������أ��õ��Ĺ�������Ϊ g��
��6����ҵ��ˮ�г����в�ͬ���͵���Ⱦ��ɲ��ò�ͬ�ķ�����������������ͬѧ��Ժ���ͬ��Ⱦ��ķ�ˮ����Ĵ�����ʩ�ͷ�����������ȷ����
|
ѡ�� |
��Ⱦ�� |
������ʩ |
������� |
|
A |
���� |
����ʯ���к� |
������ |
|
B |
Cu2+���ؽ������� |
��������� |
��ѧ�� |
|
C |
�������л���ķ�ˮ |
ͨ�������л |
������ |
|
D |
���Եķ�ˮ |
��CO2���к� |
��ѧ�� |