��Ŀ����
�����飨CH3CH2Br����һ��������ˮ���ܶ�ԼΪˮ���ܶȵ�1.5�����е�Ϊ38.4 �����ɫҺ�壬ʵ�������Ҵ���CH3CH2OH����������ķ�Ӧ���£�NaBr+H2SO4![]() NaHSO4+HBr
NaHSO4+HBr
CH3CH2OH+HBr![]() CH3CH2Br+H2O
CH3CH2Br+H2O
��֪��Ӧ���������0.3 mol NaBr(����)��0.25 mol�Ҵ���36 mLŨ���ᣨ98%���ܶ�Ϊ1.84 g��cm-3����25 mLˮ�������Ҵ����ܶ�ԼΪˮ���ܶȵ�4/5���Իش�
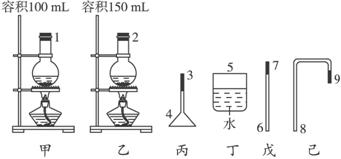
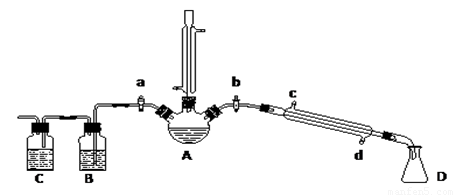
��1��������ͼ��ʾ��������װ��ȡ���ռ��������װ�ã�Ҫ��ﵽ��ȫ����ʧ�١�����Ⱦ������Ŀ�ġ��й�������ѡ�������˳��Ϊ____________�������֣���
��2��д������ѡ�õ�����������____________________��
��3����Ӧʱ���¶ȹ��ߣ����Թ۲쵽����ɫ�������ɣ�д���˷�Ӧ�Ļ�ѧ����ʽ_________
____________________��
��4�����ػ�ɫ�Ĵ���������ȡ��ɫ��������Ӧ������Լ���___________������ʹ�õ�������________��
��5����ʵ��IJ���Ϊ60%�������ȡ������________g��
��1��2 8 9 3 4 5
��2���ף��ݻ�С����Ӧ�ﳬ�����ݻ���2/3���죺����������β��������ɻ�����Ⱦ������������
��3��2HBr+H2SO4(Ũ)![]() Br2+SO2��+2H2O
Br2+SO2��+2H2O
��4��NaOH��Һ ��Һ©��
��5��16.35
��������1���ס�������Ϊ���������ȡװ�ù�ѡ�õģ�Ӧ�ÿ���ʵ����Һ�����Ӧ������ƿ�ݻ���1/3��2/3Ϊ�ˡ�0.25 mol�Ҵ������Ϊ0.25 mol��46 g��mol-1��0.8 g��cm-3��14 mL������Һ���Ϻ����ǻ��ʱҺ�������仯����V��=36 mL+25 mL+14 mL=75 mL����һ��������˼������ݻ���2/3����Ӧѡ����������
��2�����ڲ�Ʒ�������飩�ķе�ͣ�����ʱ�ᱻ���������ǵ������鲻����ˮ���ܶȱ�ˮ�ʿ���ˮ�ռ������飬��ʱ���������ˮ���²㣬ˮ��Һ�����ú��������ã�����������ӷ��������������ж������ɴ˿ɼ��ռ����������Ҫ���������Ͷ������м������ҡ�
��Ӧ�����ڸ���Ӧ�ķ����ᵼ���������������л�����������HBr���壬�����������ж�������������ȫ��HBr��������ˮ��Ϊ�����������������գ���ֹ������Ӧѡ�ñ���
��3����Ӧ�¶ȸߣ�HBr��ŨH2SO4�ᷢ��������ԭ��Ӧ������ɫ����Ϊ��������
��4��������������ػ�ɫ�����ڻ�����Br2��Ҫ��ȥBr2������NaOH��Һ���ա�
��5��n(H2SO4)=36 mL��1.84 g��mL-1��98%/98 g��mol-1��0.66 mol���ӵ�һ����Ӧʽ���жϳ�HBrΪ0.3 mol���ӵڶ�����Ӧʽ�жϳ�HBr������CH3CH2Br�����ʵ�������CH3CH2OH���ʵ���ȷ��������CH3CH2Br���۲���Ϊ0.25 mol��109 g��mol-1=27.25 g,�������ʵ�ʲ���Ϊ27.25 g��60%=16.35 g��






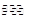 H++Cl-+HClO
H++Cl-+HClO