��Ŀ����
��һ�������£����淴ӦA2(g)��B2(g)  2C(g)��һ���ܱ������дﵽƽ��ʱ�����c(A2)=0.5mol?L-1�� c(B2)=0.1mol?L-1��c(C)=1.6mol?L-1����A2��B2��C����ʼŨ�ȷֱ�Ϊa mol?L-1��b mol?L-1��g mol?L-1��ȷ����
2C(g)��һ���ܱ������дﵽƽ��ʱ�����c(A2)=0.5mol?L-1�� c(B2)=0.1mol?L-1��c(C)=1.6mol?L-1����A2��B2��C����ʼŨ�ȷֱ�Ϊa mol?L-1��b mol?L-1��g mol?L-1��ȷ����
��1��a��gӦ����Ĺ�ϵʽΪ_________��
��2������Ӧ������Ӧ����ʼ���У���g��_________��a�����ֵΪ_________��
��3������Ӧ���淴Ӧ����ʼ���У���b��_________��a����СֵΪ_________��
��4��b��ȡֵ��ΧΪ________________��
��1��a +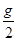 =1.3 ��2��0��1.3 ��3��0��0.4 ��4��0��b��0.9
=1.3 ��2��0��1.3 ��3��0��0.4 ��4��0��b��0.9
����

��ϰ��ϵ�д�
 ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�
�����Ŀ
 ��һ�������£����淴ӦA+B?mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯������ش��������⣺
��һ�������£����淴ӦA+B?mC�仯��ͼ��ʾ����֪�������ʾ�ڲ�ͬ�¶Ⱥ�ѹǿ��������C�ڻ�����е�����������pΪ��Ӧ��T2�¶�ʱ�ﵽƽ�����������ѹ�ı仯������ش��������⣺ 2C��g���ﵽƽ��ʱ��50%��Bת��Ϊ����C����A��ת����Ϊ25%������ʼʱ���������е�A��B�����ʵ���֮��Ϊ�� ��
2C��g���ﵽƽ��ʱ��50%��Bת��Ϊ����C����A��ת����Ϊ25%������ʼʱ���������е�A��B�����ʵ���֮��Ϊ�� �� 2C(g)�ﵽƽ��ı�־�� �� ��
2C(g)�ﵽƽ��ı�־�� �� ��