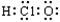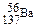��Ŀ����
���ֶ�����Ԫ�ص����ʻ�ԭ�ӽṹ��Ϣ���±���
����ݱ�����Ϣ�ش��������⣺
��1��Qԭ�ӵĵ����Ų�ʽΪ___________________________����ͬλ���� ��
��2��R��Z��ɵĻ�����Ļ�ѧʽ�� ��
��3��X��̬�⻯����ӵĿռ乹��Ϊ ��X������������Ӧ��ˮ������Ũ��Һ��ͬ����������ڻ�ѧ�����ϱ��ֵ��������� ��
��4��Y������V2O5��Ӧ�Ļ�ѧ����ʽΪ ��
��5����1.01��105Pa��298Kʱ��1.4gQR������1.6gR2��������ȫȼ�գ�����QR2����ʱ�ų�14.15kJ��������ʾQRȼ�յ��Ȼ�ѧ����ʽΪ ��
| Ԫ�� | Ԫ�����ʻ�ԭ�ӽṹ��Ϣ |
| Q | ԭ�Ӻ�����6�ֲ�ͬ�˶�״̬�ĵ��� |
| R | �����������Ǵ�����������3�� |
| X | ��̬�⻯���ˮ��Һ�������� |
| Y | ��������Ԫ�صļ����������Ӱ뾶��С |
| Z | ����Ϊ����ɫ���壬�ڿ�����ȼ�շ�����ɫ���� |
��1��Qԭ�ӵĵ����Ų�ʽΪ___________________________����ͬλ���� ��
��2��R��Z��ɵĻ�����Ļ�ѧʽ�� ��
��3��X��̬�⻯����ӵĿռ乹��Ϊ ��X������������Ӧ��ˮ������Ũ��Һ��ͬ����������ڻ�ѧ�����ϱ��ֵ��������� ��
��4��Y������V2O5��Ӧ�Ļ�ѧ����ʽΪ ��
��5����1.01��105Pa��298Kʱ��1.4gQR������1.6gR2��������ȫȼ�գ�����QR2����ʱ�ų�14.15kJ��������ʾQRȼ�յ��Ȼ�ѧ����ʽΪ ��
��1��1s22s22p2��1�֣� 12C��13C��14C ��1�֣�
��2��Na2O��Na2O2��1�֣�
��3�������Σ�1�֣����ȶ��Ժ�ǿ�����ԣ�2�֣�
 ��4��10Al+3V2O5 5Al2O3 +6V��2�֣�δ��ƽ��ȱ��Ӧ������1�֣�
��4��10Al+3V2O5 5Al2O3 +6V��2�֣�δ��ƽ��ȱ��Ӧ������1�֣�
��5��2CO(g)+O2(g)��2CO2(g)+566KJ��2��
��2��Na2O��Na2O2��1�֣�
��3�������Σ�1�֣����ȶ��Ժ�ǿ�����ԣ�2�֣�
 ��4��10Al+3V2O5 5Al2O3 +6V��2�֣�δ��ƽ��ȱ��Ӧ������1�֣�
��4��10Al+3V2O5 5Al2O3 +6V��2�֣�δ��ƽ��ȱ��Ӧ������1�֣���5��2CO(g)+O2(g)��2CO2(g)+566KJ��2��
��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ