��Ŀ����
����Ŀ����ѧ������̫���ֽܷ�ˮ���ɵ������ڴ����������������̼��Ӧ���ɼ״�����������ֱ���Լ״�Ϊȼ�ϵ�ȼ�ϵ�ء���֪H2(g)��CO(g)��CH3OH(l)��ȼ���ȷֱ�Ϊ285.8kJ��mol-1��283.0kJ��mol-1��726.5kJ��mol-1���ش��������⣺
��1����̫���ֽܷ�100gˮ���ĵ�������_______kJ��
��2���״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽΪ____________��
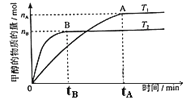
��3�����ݻ�Ϊ2L���ܱ������У���CO2��H2�ϳɼ״����������������������£������¶ȶԷ�Ӧ��Ӱ�죬ʵ������ͼ��ʾ��ע��T1��T2������300�棩������˵����ȷ����________������ţ���
���¶�ΪT1ʱ���ӷ�Ӧ��ʼ��ƽ�⣬���ɼ״���ƽ������Ϊv(CH3OH)=![]() mol��L-1��min-1
mol��L-1��min-1
�ڸ÷�Ӧ��T1ʱ��ƽ�ⳣ����T2ʱ��С
�۸÷�ӦΪ���ȷ�Ӧ
�ܴ���A��ķ�Ӧ��ϵ��T1�䵽T2���ﵽƽ��ʱ![]() ����
����
��4����T1�¶�ʱ����1molCO2��3molH2����һ�ܱպ������У���ַ�Ӧ�ﵽƽ�����CO2ת����Ϊa���������ڵ�ѹǿ����ʼѹǿ֮��Ϊ___________��
��5����ֱ���Լ״�Ϊȼ�ϵ���У��������ҺΪ���ԣ������ķ�ӦʽΪ_____________������״̬�£���ȼ�ϵ������1molCH3OH���ܲ�����������Ϊ702.1kJ�����ȼ�ϵ�ص�����Ч��Ϊ___________��ȼ�ϵ�ص�����Ч����ָ�������������������ȼ�ϵ�ط�Ӧ�����ͷŵ�ȫ������֮�ȣ���
���𰸡�1588 kJ CH3OH(l) ��O2(g) ===CO(g) + 2H2O(l) ��H��-443.5kJ��mol-1 �ۢ� ��2��a��/2 CH3OH��6e-��H2O===CO2��6H�� 96.64%
��������
��1����������ȼ���ȿ�֪ˮ�ֽ����յ�������Ȼ�����û�ѧ�������뷴Ӧ�ȵĹ�ϵ�����㣻
��2������CO��CH3OH��ȼ��������д�ȷ���ʽ�������ø�˹�����������״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽ��
��3������ͼ���м״��ı仯�����㷴Ӧ���ʣ�������ͼ����ʱ�������ʵĹ�ϵ������T1��T2��������Ӱ��ƽ����������������
��4�����ݻ�ѧƽ������η�����ƽ��ʱ�����ʵ����ʵ����������÷�Ӧǰ����������ʵ���֮�ȵ���ѹǿ֮�������
��5������ȼ�ϵ��ԭ�����������Һ���������д�缫��Ӧʽ�������������ʽ��������Ч�ʡ�
��1����H2(g)��ȼ������HΪ285.8 kJ��mol-1֪��1molH2(g)��ȫȼ������1molH2O(l)�ų�����285.8kJ�����ݻ�ѧ�������������غ�ԭ������֪�ֽ�1molH2O(l)Ϊ1molH2(g)���ĵ�����Ϊ285.8kJ����ֽ�100gH2O(l)���ĵ�����Ϊ285.8kJ��![]() =1588 kJ���ʴ�Ϊ��1588 kJ��
=1588 kJ���ʴ�Ϊ��1588 kJ��
��2����CO(g)��CH3OH(l)��ȼ������H�ֱ�Ϊ283.0 kJ��mol-1��726.5 kJ��mol-1����
��CO(g)+1/2O2(g)=CO2(g) ��H=283.0 kJ��mol-1
��CH3OH(l)+3/2O2(g)=CO2(g)+2H2O(l) ��H=726.5 kJ��mol-1
�ɸ�˹���ɿ�֪�â��ٵ÷�ӦCH3OH(l)+O2(g)=CO(g)+2H2O(l)���÷�Ӧ�ķ�Ӧ����H=726.5kJ��mol-1(283.0kJ��mol-1)=443.5kJ��mol-1���ʴ�Ϊ��CH3OH(l) ��O2(g) ===CO(g) + 2H2O(l) ��H��-443.5kJ��mol-1��
��3���������ͼ�������֪��T2�ȴﵽƽ����T2>T1�����¶����߷�Ӧ���������֪T2�ķ�Ӧ���ʴ���T1�����¶ȸ�ʱƽ��״̬CH3OH�����ʵ����٣���˵�����淴ӦCO2+3H2CH3OH+H2O���淴Ӧ�����ƶ���������ӦΪ���ȷ�Ӧ����T1ʱ��ƽ�ⳣ����T2ʱ�Ĵۡ�����ȷ�����и÷�Ӧ��T1ʱ��ƽ�ⳣ����T2ʱ�Ĵ���ڴ����а�����������ʵķ�����֪��Ӧ���ʵĵ�λ����ӦΪmol��min1����ٴ��ʴ�Ϊ���ۢܣ�
��4���ɻ�ѧƽ�������ģʽ�������֪��

������ͬ�����������ѹǿ֮�ȵ������ʵ���֮�ȣ��������ڵ�ѹǿ����ʼѹǿ֮��Ϊ![]() =(2��a)/2�� �ʴ�Ϊ��(2��a)/2��
=(2��a)/2�� �ʴ�Ϊ��(2��a)/2��
��5���״�ȼ�ϵ���У��״�Ϊ������ʧȥ���ӷ���������Ӧ�������Ϊ�������Բ���ΪCO2���ʵ缫��ӦʽΪ��CH3OH��6e-��H2O=CO2��6H�������ݼ״���ȼ���ȼ������غ㶨�ɷ�����1mol�״���Ϊȼ�ϵ�ص�ȼ�����ͷŵ�ȫ������Ϊ726.5kJ��������Ч��Ϊ��![]() ���ʴ�Ϊ��CH3OH��6e-��H2O=CO2��6H����96.64%��
���ʴ�Ϊ��CH3OH��6e-��H2O=CO2��6H����96.64%��

 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�