��Ŀ����
ʯ�ͻ����ŷŵķ�ˮ�г����ж��ֻ�ԭ�����ʡ���ˮ���м����Թ������ظ���ر���Һ����ַ�Ӧ��������������隣���Һ�ζ�ʣ����ظ���أ��Լ��ʯ�ͻ�����ˮ�л�ԭ�����ʵĺ�����
�� ����0��010mo1��L��1�������������Һ500 mLʱ�����ȡ��������茶���[��NH4��2Fe��SO4��2��6H2O]������Ϊ g��
�� ��������隣���Һ�ڿ����з��ú�Ũ�Ȼᷢ���仯�������ظ���ر���Һ�궨�����������Һ��ʵ��Ũ�ȡ���ȡ0��0040 mo1��L��1�ظ���ر���Һ10��00 mL���ζ�ʱ����ȥ25��00 mL�����������Һ������������隣���Һ��ʵ��Ũ��Ϊ ��K2Cr2O7�Ļ�ԭ����ΪCr3������
��ʵ�ʲ����У������Ա�ڱ궨�������������ҺŨ��ʱ��ÿ��ȡŨ��ֵΪc���ظ���ر���Һ10��0 mL������ʱֻҪ����ζ������ĵı������������Һ�������V����λmL���Ϳ�������һ���ļ���ʽֱ�Ӽ�����������������Һ��ʵ��Ũ�ȡ��ü���ʽΪ ��
�� 1��96g
�� 0��0096 mo1��L��1
�� c[��NH4��2Fe��SO4��2��6H2O] ��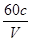 mol/L
mol/L
����������

 ����ˮ���м����Թ������ظ���ر���Һ����ַ�Ӧ��������������隣���Һ�ζ�ʣ����ظ���أ��Լ��ʯ�ͻ�����ˮ�л�ԭ�����ʵĺ�����
����ˮ���м����Թ������ظ���ر���Һ����ַ�Ӧ��������������隣���Һ�ζ�ʣ����ظ���أ��Լ��ʯ�ͻ�����ˮ�л�ԭ�����ʵĺ�����