��Ŀ����
ʵ���е����в�����ȷ���ǣ� ��
A. ���Թ�ȡ���Լ�ƿ�е�Na2CO3��Һ������ȡ�����࣬Ϊ�˲��˷ѣ��ְѹ������Լ������Լ�ƿ��
B. Ba��NO3��2����ˮ���ɽ�����Ba��NO3��2�ķ�Һ����ˮ���У�����ˮ������ˮ��
C. ����������ʹNaCl����Һ������ʱ��Ӧ����������NaCl��Һȫ����������
D. ��Ũ��������һ�����ʵ���Ũ�ȵ�ϡ����ʱ��Ũ��������ˮ��Ӧ��ȴ�����²���ת�Ƶ�����ƿ��
 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д���1����ӦFe(s)+CO2(g) FeO(s)+CO(g) ��H1��ƽ�ⳣ��ΪK1��
FeO(s)+CO(g) ��H1��ƽ�ⳣ��ΪK1��
��ӦFe(s)+H2O(g) FeO(s)+H2(g) ��H2��ƽ�ⳣ��ΪK2��
FeO(s)+H2(g) ��H2��ƽ�ⳣ��ΪK2��
�ڲ�ͬ�¶�ʱK1��K2��ֵ���±���
700�� | 900�� | |
K1 | 1��47 | 2��15 |
K2 | 2��38 | 1��67 |
�ٷ�Ӧ CO2(g) + H2(g) CO(g) + H2O(g) ��H��ƽ�ⳣ��K����H = ���æ�H1�ͦ�H2��ʾ����K= ����K1��K2��ʾ�����������������֪����ӦCO2(g) + H2(g)
CO(g) + H2O(g) ��H��ƽ�ⳣ��K����H = ���æ�H1�ͦ�H2��ʾ����K= ����K1��K2��ʾ�����������������֪����ӦCO2(g) + H2(g) CO(g) + H2O(g)�� ��Ӧ������ȡ����ȡ�����
CO(g) + H2O(g)�� ��Ӧ������ȡ����ȡ�����
��2��һ���¶��£���ij�ܱ������м����������۲�����һ������CO2���壬������ӦFe(s)+CO2(g) FeO(s)+CO(g) ��H > 0��CO2��Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��
FeO(s)+CO(g) ��H > 0��CO2��Ũ����ʱ��Ĺ�ϵ��ͼ��ʾ��
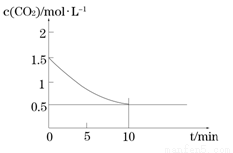
�� �������·�Ӧ��ƽ�ⳣ��Ϊ ��
�� ���д�ʩ����ʹƽ��ʱ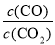 ������� ������ţ�
������� ������ţ�
A�������¶� B������ѹǿ
C������һ������CO2 D���ټ���һ��������
�� һ���¶��£���һ���̶��ݻ����ܱ������з���������Ӧ���������жϸ÷�Ӧ�ﵽ��ѧƽ��״̬���� ������ĸ����
a��������ѹǿ����
b��������ܶȲ��ٸı�
c��v��(CO2)=v��(CO)
d��c(CO2)=c(CO)
e�����������������ʵ�������


 CH3OH(g) ��H ��_______kJ��mol��1��
CH3OH(g) ��H ��_______kJ��mol��1��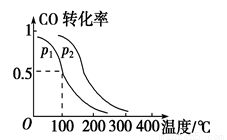
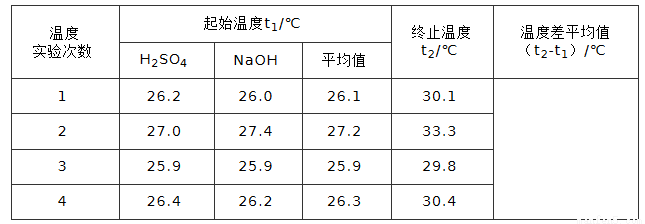
 C��g����D��g���Ѵﵽƽ��״̬��
C��g����D��g���Ѵﵽƽ��״̬��