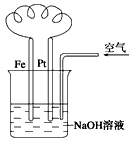
��Ŀ����
��Դ�����ǵ�ǰ����������ٵ�һ���ش���⣬H2��CO��CH3OH������Ҫ����Դ���ʣ����ǵ�ȼ��������Ϊ285.8 kJ/mol��282.5 kJ/mol��726.7 kJ/mol��
��ش�
(1)��֪CO��H2��һ�������¿��Ժϳɼ״���CO+2H2=CH3OH����H2��CO��Ӧ����CH3OH���Ȼ�ѧ����ʽΪ��________��
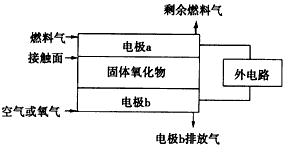
(2)��ͼΪij��ȼ�ϵ�صĹ���ԭ��ʾ��ͼ��a��b��Ϊ���Ե缫��
��ش�
(1)��֪CO��H2��һ�������¿��Ժϳɼ״���CO+2H2=CH3OH����H2��CO��Ӧ����CH3OH���Ȼ�ѧ����ʽΪ��________��
(2)��ͼΪij��ȼ�ϵ�صĹ���ԭ��ʾ��ͼ��a��b��Ϊ���Ե缫��
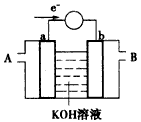
��ʹ��ʱ��������___��ͨ�루�A����B������
�ڼ���ʹ�õġ�ȼ�ϡ��Ǽ״���a ���ĵ缫��ӦʽΪ��_______________��
�ۼ���ʹ�õġ�ȼ�ϡ���ˮú�����ɷ�ΪCO��H2�����������ֵ�ص��ͭ�����ƽ�������6.4 g�����������ı�״����ˮú�������Ϊ____________��
�ڼ���ʹ�õġ�ȼ�ϡ��Ǽ״���a ���ĵ缫��ӦʽΪ��_______________��
�ۼ���ʹ�õġ�ȼ�ϡ���ˮú�����ɷ�ΪCO��H2�����������ֵ�ص��ͭ�����ƽ�������6.4 g�����������ı�״����ˮú�������Ϊ____________��
(1)CO(g)+2H2(g)=CH3OH(l) ��H=-127.4 kJ/mol
(2)��B����CH3OH-6e-+8OH-=CO32-+6H2O����2.24 L
(2)��B����CH3OH-6e-+8OH-=CO32-+6H2O����2.24 L

��ϰ��ϵ�д�
 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�
�����Ŀ
 2NH3��g������H��0
2NH3��g������H��0