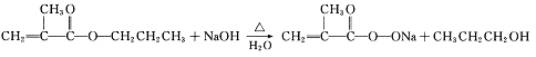��Ŀ����
(1)����֧���Ļ�����A�ķ���ʽΪC4H6O2��A����ʹBr2�����Ȼ�̼��Һ��ɫ.1molA��1molNaHCO3����ȫ��Ӧ����A�Ľṹ��ʽ��________________.д����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽ________________.
(2)������B����C��H��O����Ԫ�أ�������Ϊ60������̼����������Ϊ60���������������Ϊ13.33��.B�ڴ���Cu�������±�����������C��C�ܷ���������Ӧ����B�Ľṹ��ʽ��________________.
(3)D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����Ӧ�ķ�Ӧ��ѧ����ʽ��________________.
�𰸣�
������
������
(2)CH3CH2CH2OH (3)
|

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ