��Ŀ����
20����֪��Ӧ����2CH3OH��g���TCH3OCH3��g��+H2O��g����H1=-23.9KJ•mol-1
��2CH3OH��g���TC2H4 ��g��+2H2O��g��?��H2=-29.1KJ•mol-1
��CH3CH2OH��g���TCH3OCH3��g����H3=+50.7KJ•mol-1��
��C2H4��g��+H2O��g���TCH3CH2OH��g����H4�У���H4���ڣ�������
| A�� | -48.5 KJ•mol-1 | B�� | +48.5 KJ•mol-1 | C�� | -45.5KJ•mol-1 | D�� | +45.5 KJ•mol-1 |
���� ���ø�˹���ɹ���Ŀ���Ȼ�ѧ����ʽ�����ʱ䣬���ݸ�˹���ɢ�-��-�ۿɵã�C2H4��g��+H2O��g���TC2H5OH��g����
��� �⣺�״���ˮ��Ӧ��2CH3OH��g��=CH3OCH3��g��+H2O��g��?��H1=-23.9KJ•mol-1
�״���ϩ����Ӧ��2CH3OH��g��=C2H4 ��g��+2H2O��g��?��H2=-29.1KJ•mol-1
�Ҵ��칹����Ӧ��CH3CH2OH��g��=CH3OCH3��g����?��H3=+50.7KJ•mol-1��
���ݸ�˹���ɢ�-��-�ۿɵã�C2H4��g��+H2O��g���TC2H5OH��g����H=��-23.9+29.1-50.7��kJ/mol=-45.5kJ/mol��
�ʴ�Ϊ��-45.5 kJ•mol-1��
���� ���⿼���˹���ɵ�Ӧ�ã�Ϊ��Ƶ���㣬���ط������������������Ŀ��飬��Ŀ�ϼ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
11��Ϊ�ﵽ�±��е�ʵ��Ŀ�ģ���ѡ����ʵ��Լ���ʵ�鷽���������������Ӧ�Ŀո�
��ѡ��Ļ�ѧ�Լ���ʵ�鷽��
A������
B���μӵ�ˮ
C������KSCN��Һ
D��ͨ�����ȵ�CuO��
| ʵ��Ŀ�� | �Լ������� |
| ֤���ع����е��� | B |
| ��ȥCO2�л��е�����CO | D |
| ����FeCl2��Һ�Ƿ���� | C |
| ��ȥNa2CO3������NaHCO3 | A |
A������
B���μӵ�ˮ
C������KSCN��Һ
D��ͨ�����ȵ�CuO��
8�������й����ʵĽṹ�����ʵ������У�������ǣ�������
| A�� | ˮ����֮������ķ����Ե��±���ˮ���ӵĶѻ���ʽ�����ڷ����ܶѻ� | |
| B�� | �ɼ��Լ����ɵķ��ӿ����ǷǼ��Է��� | |
| C�� | ˮ������ˮ�����ж������ | |
| D�� | ���Ӿ�����һ�����ڷ��Ӽ������������ܴ��ڹ��ۼ� |
15�����ڵ����ʵ�����˵����ȷ���ǣ�������
| A�� | ��Ȼ�������н���C��H��O��N����Ԫ�� | |
| B�� | �ؽ�������ʹ�����ʱ��ԣ�������ʳ�ؽ����λ��ж� | |
| C�� | ���Ȼ�ʹ�����ʱ��ԣ�������Լ�������Ժ� | |
| D�� | ���������ʳ�Σ���ʹ�����ʱ��� |
5�������������������ȷ���ǣ�������
| A�� | �Ȼ���--�� | B�� | ����--�� | C�� | ������--���� | D�� | ����--������ |
9���ڱ�״���£�������������Ĺ�ϵ�У��Ӵ�С���ǣ�������
��6.72L CH4����3.01��1023��HCl���Ӣ�13.6g H2S����0.2mol NH3��
��6.72L CH4����3.01��1023��HCl���Ӣ�13.6g H2S����0.2mol NH3��
| A�� | ������ܣ��٣��ڣ��� | B�� | �ܶȣ��ڣ��ۣ��ܣ��� | ||
| C�� | �������ڣ��ۣ��٣��� | D�� | ��ԭ�������٣��ڣ��ۣ��� |
10��Ҫ�����ʵ��֤����ij���ε�ˮ�������ȵģ�����λͬѧ�ֱ�������·��������в���ȷ���ǣ�������
| A�� | ��ͬѧ���ü��ȵķ����ɳ�ȥKNO3��Һ�л��е�Fe3+��˵����Fe3+����ˮ�������ȵ� | |
| B�� | ��ͬѧ��������茶�������ˮ����ˮ���½���˵�������ˮ�������ȵ� | |
| C�� | ��ͬѧ��ͨ��ʵ�鷢��ͬŨ�ȵ��ȵĴ�����Һ����Ĵ�����Һȥ����Ч���ã�˵��̼�� ��ˮ�������ȵ� | |
| D�� | ��ͬѧ���ڴ�������Һ�е����̪��Һ�����ȣ�������ˮ������������ɫ���˵��������ˮ�������ȵ� |
 þ�����ֱ����Ũ�ȡ�������Ĺ���ϡ���ᷴӦ����������������V����ʱ�䣨t����ϵ��ͼ����Ӧ��þ����������֮��Ϊ4��3����ȫ��Ӧ��Ҫ��ʱ��֮��Ϊ2��3��
þ�����ֱ����Ũ�ȡ�������Ĺ���ϡ���ᷴӦ����������������V����ʱ�䣨t����ϵ��ͼ����Ӧ��þ����������֮��Ϊ4��3����ȫ��Ӧ��Ҫ��ʱ��֮��Ϊ2��3��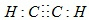 �� ��2-��-1-��ϩ�ļ���ʽ
�� ��2-��-1-��ϩ�ļ���ʽ ��
��