��Ŀ����
ijѧ����Ũ�������ʵ�ʵ�飺
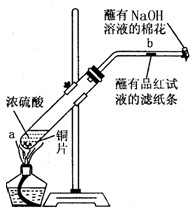
��һ֧�Թ��з���һ���С��ͭƬ���ټ���2mLŨ���ᣬȻ����Թ̶ܹ�������̨�ϡ���һС��պ��Ʒ����Һ����ֽ������е�����Ƥ���IJ������С������Թܿڣ��ڲ����ܿڴ�����һ��պ��NaOH��Һ�����������Թܣ��۲�����![]()
�ش��������⣺
��1��д���Թ��з�����Ӧ�Ļ�ѧ����ʽ ��
��2���Թ��е�Һ�巴Ӧһ��ʱ���b����ֽ���ı仯Ϊ �����Թ��з�Ӧֹͣ�������ܷ���պ��Ʒ����Һ����ֽ�����ȣ���ֽ���ı仯Ϊ ��
��3��պ��NaOH��Һ������������ ��
��4��������������γɹ��̿������з�Ӧ�е� ����ʾ��
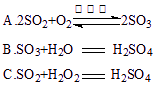 O%
O%
(1) Cu+2H2SO4(Ũ)![]() CuSO4 + SO2��+2H2O��2�֣�O%
CuSO4 + SO2��+2H2O��2�֣�O%
(2) պ��Ʒ����Һ����ֽ��ɫ��1�֣� ��ֽ��죨1�֣�
��3�����ն����SO2����ֹ��Ⱦ��������1�֣�
(4) A B ��2�֣�

��ϰ��ϵ�д�
�����Ŀ
 ijѧ����Ũ�������ʵ�ʵ�飺
ijѧ����Ũ�������ʵ�ʵ�飺

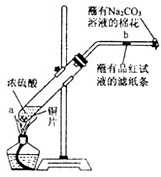 ijѧ����Ũ�������ʵ�ʵ�飺
ijѧ����Ũ�������ʵ�ʵ�飺


