��Ŀ����
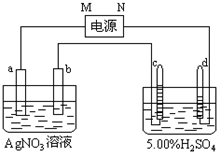 ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ�����c��d�����Ϲ��ռ���336mL����״̬�����壮�ش�
ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ�����c��d�����Ϲ��ռ���336mL����״̬�����壮�ش���1��ֱ����Դ�У�MΪ
��
��
������2��Pt�缫�����ɵ�������
��
��
��������Ϊ2.16
2.16
g����3����Դ����ĵ��ӣ������ʵ�����缫b��c��d�ֱ�����
�����ʵ����ʵ���֮��Ϊ��2��
2
2
��0.5
0.5
��1
1
����4��AgNO3��Һ��Ũ�ȣ�������С�䣮��ͬ��
����
����
��AgNO3��Һ��pH
����
����
��H2SO4��Һ��Ũ������
����
��H2SO4��Һ��pH��С
��С
����5����H2SO4��Һ������������5.00%��Ϊ5.02%����ԭ��5.00%��H2SO4��Һ������Ϊ
45.18
45.18
g����������1��5.00%��ϡH2SO4��ʵ�����ǵ�����е�ˮ����������H2����������O2������װ��ͼ�� �缫�������������c�缫��������d�缫���ɵ������������ݵ��ԭ�������жϵ缫��
��2�����ݣ�1�������ж�aΪ������bΪ�����������ǵ��װ�ã����ݵ����غ����õ���
��3�����ݵ缫��Ӧ�͵����غ����õ���
��4��װ�����ǵ��װ�ã��������ҺŨ�Ȳ��䣬��ҺPH���䣻���������Һ�ǵ��ˮ��������ҺŨ������
��5��������֪���������ǵ��ˮ���������ʲ�����ʽ���㣻
��2�����ݣ�1�������ж�aΪ������bΪ�����������ǵ��װ�ã����ݵ����غ����õ���
��3�����ݵ缫��Ӧ�͵����غ����õ���
��4��װ�����ǵ��װ�ã��������ҺŨ�Ȳ��䣬��ҺPH���䣻���������Һ�ǵ��ˮ��������ҺŨ������
��5��������֪���������ǵ��ˮ���������ʲ�����ʽ���㣻
����⣺��1��5.00%��ϡH2SO4��ʵ�����ǵ�����е�ˮ����������H2����������O2����V��H2����V��O2��=2��1������װ��ͼ�� �缫���������������ȷ��d��Ϊ���������Դ��N��Ϊ������c��Ϊ������bΪ������aΪ������MΪ�������ʴ�Ϊ������
��2������������Һ���ˮ�ķ�Ӧ��V��H2��=336mL��2/3=224mL����Ϊ0.01mol��V��O2��=336mL��1/3=112mL����Ϊ0.005mol��˵����·����0.02mol���ӣ������b����Pt������������Ag��0.02��108=2.16g��
�ʴ�Ϊ������2.16g��
��3����Դ����ĵ��ӣ������ʵ�����缫b��c��d�ֱ����ɵ����ʵ����ʵ���֮��Ϊ��n��e����n��Ag����n��O2����n��H2��=0.02��0.02��0.005��0.01=2��2��1/2��1��
�ʴ�Ϊ��2��0.5��1��
��4����Ag�������缫��Pt�������缫��AgNO3��Һ��ɵĵ�Ƴأ����AgNO3��ҺŨ�Ȳ��䣬pHҲ���䣮���5.00%��H2SO4��Һ���������е�ˮ������⣬���H2SO4��ҺŨ������pH��С��
�ʴ�Ϊ�����䣬���䣬����С��
��5����ԭ5.00%��H2SO4��ҺΪxg���������ˮ0.01��18=0.18g����5.00%x=5.02%��x-0.18������ã�x=45.18g��
�ʴ�Ϊ��45.18��
��2������������Һ���ˮ�ķ�Ӧ��V��H2��=336mL��2/3=224mL����Ϊ0.01mol��V��O2��=336mL��1/3=112mL����Ϊ0.005mol��˵����·����0.02mol���ӣ������b����Pt������������Ag��0.02��108=2.16g��
�ʴ�Ϊ������2.16g��
��3����Դ����ĵ��ӣ������ʵ�����缫b��c��d�ֱ����ɵ����ʵ����ʵ���֮��Ϊ��n��e����n��Ag����n��O2����n��H2��=0.02��0.02��0.005��0.01=2��2��1/2��1��
�ʴ�Ϊ��2��0.5��1��
��4����Ag�������缫��Pt�������缫��AgNO3��Һ��ɵĵ�Ƴأ����AgNO3��ҺŨ�Ȳ��䣬pHҲ���䣮���5.00%��H2SO4��Һ���������е�ˮ������⣬���H2SO4��ҺŨ������pH��С��
�ʴ�Ϊ�����䣬���䣬����С��
��5����ԭ5.00%��H2SO4��ҺΪxg���������ˮ0.01��18=0.18g����5.00%x=5.02%��x-0.18������ã�x=45.18g��
�ʴ�Ϊ��45.18��
���������⿼����ԭ����Ӧ�ã���Ҫ�ǵ������е����غ�ļ���Ӧ�ã���������缫�жϷ�������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�
�����Ŀ
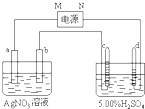 ��ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ���ֻ��c��d�����Ϲ��ռ���336mL����״̬�����壮�ش�
��ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ���ֻ��c��d�����Ϲ��ռ���336mL����״̬�����壮�ش�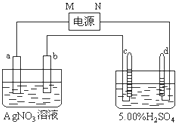 ��ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ���b�缫��֣�����c��d�缫����������������Ϲ��ռ���336mL����״̬�����壮�ش�
��ͼ�е缫a��b�ֱ�ΪAg�缫��Pt�缫���缫c��d����ʯī�缫��ͨ��һ��ʱ���b�缫��֣�����c��d�缫����������������Ϲ��ռ���336mL����״̬�����壮�ش�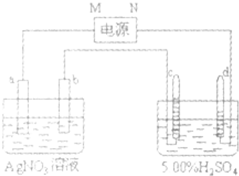 ͼ�е缫a��b�ֱ�ΪAg�缫��Pt������缫c��d����ʯī�缫��ͨ��һ��ʱ�����c��d�������ռ���336mL����״�������壬�������Ϊ1��2���ش�
ͼ�е缫a��b�ֱ�ΪAg�缫��Pt������缫c��d����ʯī�缫��ͨ��һ��ʱ�����c��d�������ռ���336mL����״�������壬�������Ϊ1��2���ش�