��Ŀ����
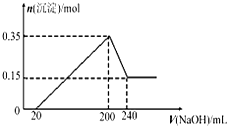
��һ��������Mg��Al�Ļ����Ͷ��500 mLϡ�����У�����ȫ���ܽⲢ�������塣����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��

������˵����ȷ����
A��Mg��Al��������Ϊ8 g
B����������ʵ���Ũ��Ϊ5 mol��L��1
C��NaOH��Һ�����ʵ���Ũ��Ϊ5 mol��L��1
D�����ɵ�H2�ڱ�״���µ����Ϊ11.2 L
C��
����:
��ͼ���֪��þ�����ʵ���Ϊ0.15 mol���������ʵ���Ϊ0.2 mol������������Ϊ��27 g��mol��1��0.2 mol��24 g��mol��1��0.15 mol��9.0 g��A�����������������Ϊ��(0.2��1.5��0.15) mol��22.4 L��mol��1<11.2 L�����D�����þ���Ӻ���������ȫ�������������Ƶ����ʵ���Ϊ��0.15 mol��2��0.2 mol��3��0.9 mol�����������Ƶ�Ũ��Ϊ��0.9 mol��0.18 L��5 mol��L��1��C����ȷ���������������ʱ����Һ������Ϊ�����ƣ�����������غ�������Ũ��Ϊ��0.2 L��5 mol��L��1��2��0.5 L��1 mol��L��1�����B�����

��ϰ��ϵ�д�
�����Ŀ
 ��һ��������Mg��Al�Ļ����Ͷ��500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵������ȷ���ǣ�������
��һ��������Mg��Al�Ļ����Ͷ��500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵������ȷ���ǣ�������| A��Mg��Al��������Ϊ9g | B�����ɵ�H2�ڱ�״���µ����Ϊ11.2L | C��NaOH��Һ�����ʵ���Ũ��Ϊ5mol/L | D����������ʵ���Ũ��Ϊ1mol/L |
 ��һ��������Mg��Al�Ļ����Ͷ��250mL��2.0mol/Lϡ�����У�����ȫ���ܽⲢ��������0����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ�������
��һ��������Mg��Al�Ļ����Ͷ��250mL��2.0mol/Lϡ�����У�����ȫ���ܽⲢ��������0����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ������� ��2009?����һģ����һ��������Mg��Al�Ļ����Ͷ��1mol?L-1500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ�������
��2009?����һģ����һ��������Mg��Al�Ļ����Ͷ��1mol?L-1500mLϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ������� ��һ��������Mg��Al�����Ͷ��500mL ϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ�������
��һ��������Mg��Al�����Ͷ��500mL ϡ�����У�����ȫ���ܽⲢ�������壮����Ӧ��ȫ����������Һ�м���NaOH��Һ�����ɳ��������ʵ��������NaOH��Һ�������ϵ��ͼ��ʾ��������˵����ȷ���ǣ�������