��Ŀ����
13��X��Y��L��MΪ�˵�������������ǰ20������Ԫ�أ�X2����������壬Y��L��M����Ԫ�ص���������Ϊ5�ı������ش��������⣺��1��X��L��ɵ��������ĵ���ʽΪ
 ��
����2��X��M��ɵ�����Ϊ���ӣ�����ۡ������ӡ�������������ʿ���ΪҰ�����Ӧ��ȼ�ϣ�����ˮ��Ӧ�Ļ�ѧ����ʽΪCaH2+2H2O�TCa��OH��2+2H2����
��3��Y�����ڱ��е�λ���ǵڶ����ڢ�A�壬�京����Ļ�ѧʽΪH3BO3��
��4��L��M�γɵĻ�������L�Ļ��ϼ���-3��
���� X2����������壬XӦΪHԪ�أ�Y��L��M����Ԫ�ص���������Ϊ5�ı�������Ϊ����Ԫ�أ����������ֱ�Ϊ5��15��20���ֱ�B��P��Ca��Ԫ�أ���϶�Ӧ���ʡ�������������Լ�Ԫ�������ʽ����⣮
��� �⣺��1��X��L��ɵ��������ΪPH3������ʽ���ư�����Ϊ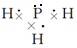 ���ʴ�Ϊ��
���ʴ�Ϊ��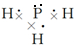 ��
��
��2��X��M��ɵ�����ΪCaH2��Ϊ���ӻ��������ˮ��Ӧ��������������ʽΪCaH2+2H2O�TCa��OH��2+2H2����
�ʴ�Ϊ�����ӣ�CaH2+2H2O�TCa��OH��2+2H2����
��3��YΪBԪ�أ�λ�����ڱ��ڶ����ڢ�A�壬������ΪH3BO3���ʴ�Ϊ���ڶ����ڢ�A�壻H3BO3��
��4��L��M�γɵĻ�����Ca3P2��Ca�Ļ��ϼ�Ϊ+2�ۣ�PΪ-3�ۣ��ʴ�Ϊ��-3��
���� ���⿼��ԭ�ӽṹ��Ԫ�������ʣ�Ϊ��Ƶ���㣬���ؿ���ѧ���ķ���������ע����ճ���Ԫ�������ڱ���λ�������ʵĵݱ���ɣ��ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
3������ʵ�����������ͽ��۾���ȷ���ǣ�������
| ѡ�� | ʵ�� | ���� | ���� |
| A | ��Ũ������̼��Ӧ���ɵ���������ͨ����������ˮ�ͳ���ʯ��ˮ | ��ˮ��ɫ������ʯ��ˮ����� | ����ΪSO2��CO2�Ļ���� |
| B | ����AgI��������Һ�е���Ũ�� KC1��Һ | �а�ɫ�������� | AgCl��Ksp��AgI��KspС |
| C | ������Һ������۵ķ�Ӧװ�������� ��ʢ��CCl4��AgNO3��Һ��ϴ��ƿ | AgNO3��Һ�г��ֵ���ɫ���� | ����Һ��Ӧ�巢����ȡ�� |
| D | ��NaI��FeCl3��Һ���Թ��л�Ϻ��뱽�������� | �²���Ϻ�ɫ | �����ԣ�Fe3+��I2 |
| A�� | A | B�� | B | C�� | C | D�� | D |
4����NAΪ�����ӵ�������ֵ������������ȷ���ǣ�������
| A�� | 1molCl2������Fe��Ӧת�Ƶ�����һ��Ϊ3NA | |
| B�� | ��״���£�2.24LNH3�к��й��ۼ�����ĿΪNA | |
| C�� | ����£�11.2LSO3�����ķ�����ĿΪ0.5NA | |
| D�� | lmolNa2O��Na2O2�����������������������3NA |
1��NA���������ӵ�������ֵ������������ȷ���ǣ�������
| A�� | 0.1 mol�İ��ף�P4��������������Ĺ��ۼ�����Ϊ0.4NA | |
| B�� | ��1 mol Cl2ͨ��ˮ�У�HC1O��Cl-��ClO-������֮��Ϊ2NA | |
| C�� | 6.4 g��S2��S4��S8��ɵĻ���ﺬ��ԭ����Ϊ0.2NA | |
| D�� | �ܱ�������2 mol NO��1 mol O2��ַ�Ӧ����ķ�����ΪNA |
8�������£���ijŨ�ȵ���ˮ�е���Na2SO3��Һ��pH�仯��ͼ��ʾ����������Ũ�ȵĹ�ϵ������ǣ�������

| A�� | a�㣺c��H+����c��Cl-����c��ClO-�� | |
| B�� | b�㣺$\frac{1}{2}$ c��Cl-���Tc��SO42-��=0.005mol•L-1 | |
| C�� | c�㣺c��Na+���T2c��HSO3-��+2c��SO32-�� | |
| D�� | d �㣺c��Na+���Tc��Cl-��+c��HSO3-��+2c��SO32-��+2c��SO42-�� |
2��NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A�� | ���³�ѹ�£�4 g H2���еķ�����Ϊ 2NA | |
| B�� | 8 g H2O�ڱ�״���µ������22.4 L | |
| C�� | 2.3 g�����Ʊ�Ϊ������ʱʧȥ�ĵ�����ΪNA | |
| D�� | ���ʵ���Ũ��Ϊ0.5 mol/L��MgCl2��Һ�У�����Cl-����ΪNA |
9����������������֬���ǣ�������
| A�� | ���� | B�� | ������ | C�� | ú�� | D�� | ���� |
6��NA��������٤��������ֵ������˵����ȷ���ǣ�������
| A�� | ����£�ij���ý���ת��2NA������ʱ�����������ᷴӦ����44.8LH2 | |
| B�� | ��Na2O2�Ƶ�2.24L����µ�O2��ת�Ƶĵ�����ĿΪ0.4NA | |
| C�� | ��98gH2SO4����ˮ��ȫ���룬����ѵĹ��ۼ���ĿΪ2NA | |
| D�� | ���³�ѹ�£�0.1molCl2����ˮ��ת�Ƶĵ�����Ϊ0.1NA |