��Ŀ����
��1����6mol H2��3mol CO�����ݻ�Ϊ0.5L���ܱ������У��������·�Ӧ��2H2������+CO������ CH3OH��������6s��ϵ��ƽ�⣬������ѹǿΪ��ʼʱ��0.6��������v��H2��=
CH3OH��������6s��ϵ��ƽ�⣬������ѹǿΪ��ʼʱ��0.6��������v��H2��=
��2����50mL HNO3��HCl�Ļ����Һ�У�����������AgNO3��Һ�������ɵij������ˡ�ϴ�ӡ�����Ƶ�������Ϊ2.87g������Һϡ����500mL�������Һ��pHΪ1��
����ԭ��Һ���������������ʵ���Ũ�ȷֱ��Ƕ��٣�
���к�ԭ�������Һ����ҪpHΪ13��Ba��OH��2��Һ�����Ϊ����mL��
 CH3OH��������6s��ϵ��ƽ�⣬������ѹǿΪ��ʼʱ��0.6��������v��H2��=
CH3OH��������6s��ϵ��ƽ�⣬������ѹǿΪ��ʼʱ��0.6��������v��H2��=1.2mol?L-1?S-1
1.2mol?L-1?S-1
��CO��ת����=60%
60%
��2����50mL HNO3��HCl�Ļ����Һ�У�����������AgNO3��Һ�������ɵij������ˡ�ϴ�ӡ�����Ƶ�������Ϊ2.87g������Һϡ����500mL�������Һ��pHΪ1��
����ԭ��Һ���������������ʵ���Ũ�ȷֱ��Ƕ��٣�
���к�ԭ�������Һ����ҪpHΪ13��Ba��OH��2��Һ�����Ϊ����mL��
��������1������ѹǿ֮�ȵ������ʵ���֮�ȣ���Ϸ���ʽ��������ʽ�����������������
��2�����ݷ�Ӧ����ط���ʽ���㣬��Ӧ�ķ���ʽ�У�HCl+AgNO3=AgCl��+HNO3��H++OH-=H2O��
��2�����ݷ�Ӧ����ط���ʽ���㣬��Ӧ�ķ���ʽ�У�HCl+AgNO3=AgCl��+HNO3��H++OH-=H2O��
����⣺��1����ƽ��ʱת����CO�����ʵ���Ϊn����
2H2������+CO������ CH3OH������
CH3OH������
��ʼ��6mol 3mol 0
ת����2n n n
ƽ�⣺��6mol-2n�� ��3mol-n�� n
��Ӧ������������ʵ���Ϊ��6mol-2n��+��3mol-n��+n=9mol-2n
ѹǿ֮�ȵ������ʵ���֮�ȣ�
��
=0.6��n=1.8mol����
��v��H2��=
=1.2mol?L-1?s-1��
�ʴ�Ϊ��1.2mol?L-1?s-1��
��CO��ת����Ϊ
��100%=60%��
�ʴ�Ϊ��60%��
��2����������2.87g AgCl������Ҫ�����ʵ���Ϊx��
HCl+AgNO3=AgCl��+HNO3
1mol 143.5g
x 2.87g
x=0.02mol
����c��HCl��=0.02mol��0.05L=0.4mol/L��
��ҺpH=1����c��H+��=0.1mol/L��
ԭ��Һ�к���H+�����ʵ���Ϊ��n��H+��=0.5L��0.1mol/L=0.05mol��
n��HNO3��=0.05mol-0.02mol=0.03mol��
c��HNO3��=
=0.6mol/L��
��ԭ��Һ���������������ʵ���Ũ�ȷֱ���0.6mol/L��0.4mol/L��
�ڷ�Ӧ����Һ��n��H+��=0.05mol
pHΪ13��Ba��OH��2��Һ�У�c��OH-��=0.1mol/L��
�跴Ӧ���ĵ�OH-�����ʵ���Ϊy
H++OH-=H2O
1mol 1mol
0.05mol y
y=0.05mol��
��Ba��OH��2��Һ�����Ϊ
=0.5L=500ml��
����ҪpHΪ13��Ba��OH��2��Һ�����Ϊ500ml��
2H2������+CO������
 CH3OH������
CH3OH��������ʼ��6mol 3mol 0
ת����2n n n
ƽ�⣺��6mol-2n�� ��3mol-n�� n
��Ӧ������������ʵ���Ϊ��6mol-2n��+��3mol-n��+n=9mol-2n
ѹǿ֮�ȵ������ʵ���֮�ȣ�
��
| 9mol-2n |
| 9mol |
��v��H2��=
| ||
| 6s |
�ʴ�Ϊ��1.2mol?L-1?s-1��
��CO��ת����Ϊ
| 1.8mol |
| 3mol |
�ʴ�Ϊ��60%��
��2����������2.87g AgCl������Ҫ�����ʵ���Ϊx��
HCl+AgNO3=AgCl��+HNO3
1mol 143.5g
x 2.87g
x=0.02mol
����c��HCl��=0.02mol��0.05L=0.4mol/L��
��ҺpH=1����c��H+��=0.1mol/L��
ԭ��Һ�к���H+�����ʵ���Ϊ��n��H+��=0.5L��0.1mol/L=0.05mol��
n��HNO3��=0.05mol-0.02mol=0.03mol��
c��HNO3��=
| 0.03mol |
| 0.05L |
��ԭ��Һ���������������ʵ���Ũ�ȷֱ���0.6mol/L��0.4mol/L��
�ڷ�Ӧ����Һ��n��H+��=0.05mol
pHΪ13��Ba��OH��2��Һ�У�c��OH-��=0.1mol/L��
�跴Ӧ���ĵ�OH-�����ʵ���Ϊy
H++OH-=H2O
1mol 1mol
0.05mol y
y=0.05mol��
��Ba��OH��2��Һ�����Ϊ
| 0.05mol |
| 0.1mol/L |
����ҪpHΪ13��Ba��OH��2��Һ�����Ϊ500ml��
���������⿼�黯ѧƽ���Լ�����ʽ�ļ��㣬��Ŀ�Ѷ��еȣ�ע�����ʵ���Ӧ���ڻ�ѧ����ʽ�ļ��㣮

��ϰ��ϵ�д�
 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д� �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д�
�����Ŀ
 ��2014?����һģ���̶�������CO2������Ч��������Դ�������ٿ����е��������壮��ҵ�������о�����CO2�������״�ȼ�ϵķ������÷����Ļ�ѧ����ʽ�ǣ�
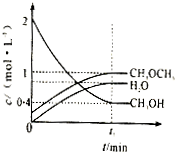
��2014?����һģ���̶�������CO2������Ч��������Դ�������ٿ����е��������壮��ҵ�������о�����CO2�������״�ȼ�ϵķ������÷����Ļ�ѧ����ʽ�ǣ� ��2012?��������ģ��T��ʱ����6mol CO2��8mol H2����2L�ܱ������У�������ӦCO2��g��+3H2��g��?CH3OH��g��+H2O��g����������H2�����ʵ�����ʱ��仯��ͼ��ʵ����ʾ��ͼ�����߱�ʾ���ı�ijһ��Ӧ����ʱ��H2�����ʵ�����ʱ��ı仯������˵����ȷ���ǣ�������
��2012?��������ģ��T��ʱ����6mol CO2��8mol H2����2L�ܱ������У�������ӦCO2��g��+3H2��g��?CH3OH��g��+H2O��g����������H2�����ʵ�����ʱ��仯��ͼ��ʵ����ʾ��ͼ�����߱�ʾ���ı�ijһ��Ӧ����ʱ��H2�����ʵ�����ʱ��ı仯������˵����ȷ���ǣ�������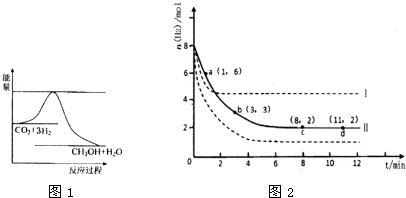
 ��1������ˮú���ϳɶ����ѣ�CH3OCH3�����Ȼ�ѧ����ʽΪ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g������H=-274KJ/mol���÷�Ӧ��һ�������µ��ܱ������дﵽƽ���Ϊͬʱ��߷�Ӧ���ʺͶ����ѵIJ��ʣ����Բ�ȡ�Ĵ�ʩ��
��1������ˮú���ϳɶ����ѣ�CH3OCH3�����Ȼ�ѧ����ʽΪ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g������H=-274KJ/mol���÷�Ӧ��һ�������µ��ܱ������дﵽƽ���Ϊͬʱ��߷�Ӧ���ʺͶ����ѵIJ��ʣ����Բ�ȡ�Ĵ�ʩ��