��Ŀ����
4������˵�����ʾ��������ȷ���ǣ�������| A�� | ����ͬһ����ѧ��Ӧ��������һ����ɻ��Ƿּ�����ɣ��䷴Ӧ���ʱ���ͬ | |
| B�� | ����������������̬���ʷֱ���ȫȼ�գ����߷ų��������� | |
| C�� | ��ʾ����ȼ���ȵġ�HΪ-285.8 kJ•mol-1��������ȼ�յ��Ȼ�ѧ����ʽΪ��2H2��g��+O2��g���T2H2O��l����H=-285.8 kJ•mol-1 | |
| D�� | ��֪�к��ȡ�HΪ-57.3 kJ•mol-1��������0.5 mol H2SO4��Ũ��Һ�뺬1 mol NaOH����Һ��ϣ��ų�����������57.3 kJ |
���� A��������һ����ɻ��Ƿּ�����ɣ�ʼ��̬��ͬ��
B���������ȹ�̬�������ߣ�
C��ȼ����Ϊ1mol����ȼ�������ȶ�������������仯��
D������1molˮ�ų�����Ϊ57.3kJ��Ũ����ϡ�ͷ��ȣ�
��� �⣺A��������һ����ɻ��Ƿּ�����ɣ�ʼ��̬��ͬ����Ӧ���ʱ���ͬ����A��ȷ��
B���������ȹ�̬�������ߣ������������������̬���ʷֱ���ȫȼ�գ�ǰ�߷ų��������࣬��B����
C��ȼ����Ϊ1mol����ȼ�������ȶ�������������仯��������ȼ���ȵġ�HΪ-285.8 kJ•mol-1��������ȼ�յ��Ȼ�ѧ����ʽΪH2��g��+$\frac{1}{2}$O2��g���TH2O��l����H=-285.8 kJ•mol-1����C����
D������1molˮ�ų�����Ϊ57.3kJ��Ũ����ϡ�ͷ��ȣ���0.5 mol H2SO4��Ũ��Һ�뺬1 mol NaOH����Һ��ϣ��ų�����������57.3 kJ����D����
��ѡA��
���� ���⿼�鷴Ӧ�����ʱ䣬Ϊ��Ƶ���㣬���շ�Ӧ�������仯��ȼ�������к���Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע��ѡ��DΪ�����״��㣬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�
�����Ŀ
16����NAΪ�����ӵ���������ֵ������˵����ȷ���ǣ�������
| A�� | 0.1 mol���к��е�̼̼˫����ĿΪ0.3 NA | |
| B�� | 3.2g�����к��е���ԭ����Ϊ0.2NA | |
| C�� | 1L0.1 mol•L-1 CH3COOH��Һ�к��е���ԭ����Ϊ0.4NA | |
| D�� | ��״���£�22.4 L�����к��е�HCl������ΪNA |
15��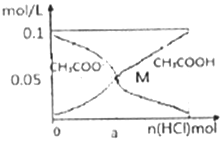 ��������l0mL0.1mol•L-1CH3COONa��Һ�У�����ͨ��HCl�����CH3COO-��CH3COOHŨ�ȵı仯������ͼ��ʾ����������Һ�ݻ��仯��������˵������ȷ���ǣ�������
��������l0mL0.1mol•L-1CH3COONa��Һ�У�����ͨ��HCl�����CH3COO-��CH3COOHŨ�ȵı仯������ͼ��ʾ����������Һ�ݻ��仯��������˵������ȷ���ǣ�������
 ��������l0mL0.1mol•L-1CH3COONa��Һ�У�����ͨ��HCl�����CH3COO-��CH3COOHŨ�ȵı仯������ͼ��ʾ����������Һ�ݻ��仯��������˵������ȷ���ǣ�������
��������l0mL0.1mol•L-1CH3COONa��Һ�У�����ͨ��HCl�����CH3COO-��CH3COOHŨ�ȵı仯������ͼ��ʾ����������Һ�ݻ��仯��������˵������ȷ���ǣ�������| A�� | ��n��HCl��=1.0��10-3molʱ����Һ�� c��Na+��=c��Cl-����c��H+����c��CH3COO-����c��OH-�� | |
| B�� | M����Һ��ˮ�ĵ���̶ȱ�ԭ��ҺС | |
| C�� | ����HCl��ͨ�룬c��OH-��/c��CH3COO-�� ֵ���ϼ�С | |
| D�� | ��M ��ʱ��c��H+��-c��OH-��=��a-0.05��mol/L |
12�����й����л��������ϵ�������У���ȫ��ȷ��һ���ǣ�������
| ѡ�� | ԭ�� | ���� |
| A | ��ϩ�ͱ�����ʹ��ˮ��ɫ | ������ϩ���Ӷ�����̼̼˫�� |
| B | ������������ϩһ�������¶��ܺ�ˮ��Ӧ | ��������ͬһ���͵ķ�Ӧ |
| C | ���ᡢ�����Ƕ��������Ƶ�������ͭ��Ӧ | ��ͬ����������Ӧ |
| D | ��ϩ��ʹ������Ȼ�̼��Һ�����Ը��������Һ��ɫ | ���ߵ���ɫ�����Dz���ͬ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
19������˵��������ǣ�������
| A�� | ����ʽ��ͬ�ģ���Ԫ�ذٷֺ���Ҳ��ͬ��������ͬ�����ʣ� | |
| B�� | ͬϵ��֮��������ƵĻ�ѧ���ʣ� | |
| C�� | ��ϩ�����㽶��ˮ���Ĵ������ | |
| D�� | ��ϩȼ��ʱ������������ͬʱ�������̣� |
9������֤�����������������ʵ�ǣ�������
| A�� | ��������Ӧ | B�� | ��Ư���� | ||
| C�� | ����������̼�ᷴӦ�д��������� | D�� | �����ֽܷ� |
16��ͬ���칹���������л���ѧ���Ƿdz��ձ�ģ������л��ﻥΪͬ���칹����ǣ�������
��CH2=CHCH3
��
��CH3CH2CH3
��HC CCH3
��
��CH3CH=CHCH3��
��CH2=CHCH3
��

��CH3CH2CH3
��HC CCH3
��

��CH3CH=CHCH3��
| A�� | �ٺ͢� | B�� | �ٺ͢� | C�� | �ٺ͢� | D�� | �ݺ͢� |
13�� ��֪����OH=-lgc��OH-����298Kʱ����20.00mL 0.10mol•L-1��ˮ�е���0.10mol•L-1�����ᣬ��Һ��pH��pOH��������������ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
��֪����OH=-lgc��OH-����298Kʱ����20.00mL 0.10mol•L-1��ˮ�е���0.10mol•L-1�����ᣬ��Һ��pH��pOH��������������ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
 ��֪����OH=-lgc��OH-����298Kʱ����20.00mL 0.10mol•L-1��ˮ�е���0.10mol•L-1�����ᣬ��Һ��pH��pOH��������������ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
��֪����OH=-lgc��OH-����298Kʱ����20.00mL 0.10mol•L-1��ˮ�е���0.10mol•L-1�����ᣬ��Һ��pH��pOH��������������ϵ��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | ����PJN��ʾ��Һ��pH�ı仯 | |
| B�� | pH��pOH�����J��Ӧ��V�����ᣩ=20.00 mL | |
| C�� | M��ˮ�ĵ���̶ȱ�Q��� | |
| D�� | M��P�������ֵ֮��a+b=14 |
3�� H2C2O4Ϊ��Ԫ���ᣮ20��ʱ����0.100mol•L-1��H2C2O4��Һ����������NaOH���壨��������Һ����仯������Һ�в����������ʵ���Ũ����pH�ı仯��ͼ��ʾ��������ָ����Һ�������ʵ���Ũ�ȵĹ�ϵһ����ȷ���ǣ�������
H2C2O4Ϊ��Ԫ���ᣮ20��ʱ����0.100mol•L-1��H2C2O4��Һ����������NaOH���壨��������Һ����仯������Һ�в����������ʵ���Ũ����pH�ı仯��ͼ��ʾ��������ָ����Һ�������ʵ���Ũ�ȵĹ�ϵһ����ȷ���ǣ�������
 H2C2O4Ϊ��Ԫ���ᣮ20��ʱ����0.100mol•L-1��H2C2O4��Һ����������NaOH���壨��������Һ����仯������Һ�в����������ʵ���Ũ����pH�ı仯��ͼ��ʾ��������ָ����Һ�������ʵ���Ũ�ȵĹ�ϵһ����ȷ���ǣ�������
H2C2O4Ϊ��Ԫ���ᣮ20��ʱ����0.100mol•L-1��H2C2O4��Һ����������NaOH���壨��������Һ����仯������Һ�в����������ʵ���Ũ����pH�ı仯��ͼ��ʾ��������ָ����Һ�������ʵ���Ũ�ȵĹ�ϵһ����ȷ���ǣ�������| A�� | Na2C2O4��Һ�У�2c��C2O42-����c��Na+�� | |
| B�� | pH=5����Һ�У�c��Na+��+c��H+��=c��OH-��+c��HC2O4-��+c��C2O42-�� | |
| C�� | c��HC2O4-��=c��C2O42-������Һ�У�c��Na+����0.100mol•L-1+c��HC2O4-�� | |
| D�� | c��Na+��=0.100mol•L-1����Һ�У�c��H+��-c��OH-��=c��C2O42-��-c��H2C2O4�� |