��Ŀ����
ij�С���÷�ͭ��ȡ����ͭ��Һ��ʵ�����Ϊ��
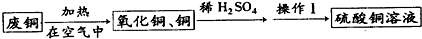
��1����ͭ�ڿ����м��Ⱥ�õ�������ͭ�к�������ͭ��ԭ����_������ĸ��ţ���
a������ʱ����ͭ�ֽ�����ͭ b��ͭ������û����ȫ��Ӧ c������ʱͭ������������Ӧ
��2������ͭ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ ��
��3��������������� ���ò����õ��IJ��������У����������ձ��� ��
��4��ʢ������ͭ��Һ����������������ԭ���ǣ��û�ѧ����ʽ���ͣ� ��
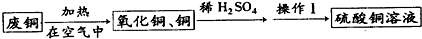
��1����ͭ�ڿ����м��Ⱥ�õ�������ͭ�к�������ͭ��ԭ����_������ĸ��ţ���
a������ʱ����ͭ�ֽ�����ͭ b��ͭ������û����ȫ��Ӧ c������ʱͭ������������Ӧ
��2������ͭ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ
��3���������������
��4��ʢ������ͭ��Һ����������������ԭ���ǣ��û�ѧ����ʽ���ͣ�
��������1����������巴Ӧ����Ϊ�Ӵ����С��ʹ��Ӧ������ȫ���У�
��2����д���ֽⷴӦ�Ļ�ѧ����ʽʱҪ�����ж��ܷ�Ӧ��Ȼ����ȷ��д��Ӧ���������Ļ�ѧʽ����ƽ��ע��������
��3�������Ƿ�����壨ͭ����Һ�壨����ͭ��Һ��������һ�ַ��������˲����õ�����Ҫ����Ҫ��ס���ر��Dz������������ձ�����������©����
��4��ʢ������ͭ��Һ������������������Ϊ��������ͭ��Һ��Ӧ��
��2����д���ֽⷴӦ�Ļ�ѧ����ʽʱҪ�����ж��ܷ�Ӧ��Ȼ����ȷ��д��Ӧ���������Ļ�ѧʽ����ƽ��ע��������
��3�������Ƿ�����壨ͭ����Һ�壨����ͭ��Һ��������һ�ַ��������˲����õ�����Ҫ����Ҫ��ס���ر��Dz������������ձ�����������©����
��4��ʢ������ͭ��Һ������������������Ϊ��������ͭ��Һ��Ӧ��
����⣺��1��ͭ�ڿ����м���ʱ�����ɵ�����ͭ�Ḳ����ͭ�ı��棬ʹͭ���ܺ������Ӵ���������ȫ��Ӧ��
�ʴ�Ϊ��b��
��2������ͭ�Ǽ���������ܺ��ᷴӦ�����κ�ˮ��������ͭ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��
CuO+H2SO4=CuSO4+H2O���ʴ�Ϊ��CuO+H2SO4=CuSO4+H2O��
��3��������壨ͭ����Һ�壨����ͭ��Һ���IJ�����������ǹ��ˣ��ò����õ��������������ձ�����������©�����ʴ�Ϊ�����ˣ�©����
��4����Ϊ����ͭ���ã���������ͭ��Һ��Ӧ����ѧ����ʽΪ��Fe+CuSO4�TFeSO4+Cu��
�ʴ�Ϊ��Fe+CuSO4�TFeSO4+Cu��
�ʴ�Ϊ��b��
��2������ͭ�Ǽ���������ܺ��ᷴӦ�����κ�ˮ��������ͭ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��
CuO+H2SO4=CuSO4+H2O���ʴ�Ϊ��CuO+H2SO4=CuSO4+H2O��
��3��������壨ͭ����Һ�壨����ͭ��Һ���IJ�����������ǹ��ˣ��ò����õ��������������ձ�����������©�����ʴ�Ϊ�����ˣ�©����
��4����Ϊ����ͭ���ã���������ͭ��Һ��Ӧ����ѧ����ʽΪ��Fe+CuSO4�TFeSO4+Cu��
�ʴ�Ϊ��Fe+CuSO4�TFeSO4+Cu��
���������⿼����ͬѧ�ǵ������������������д��ѧ����ʽ�����������Ը��ݽ̲�֪ʶ���ش��Ѷ��еȣ�

��ϰ��ϵ�д�
 �¿α�������������ҵ�������γ�����ϵ�д�
�¿α�������������ҵ�������γ�����ϵ�д� ����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
����ν����Ž̲��㽭���̴�ѧ������ϵ�д�
�����Ŀ
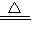 2CuO��CuO+H2SO4=CuSO4+H2O��
2CuO��CuO+H2SO4=CuSO4+H2O��