��Ŀ����
��Ԫ�ؿ��γɶ������ӣ���N3-��N3-��NH2-��NH4+��N2H5+��N2H62+�ȣ���1����N���ڵ�����Ԫ��C��O�����ߵ�һ�������ɴ�СΪ______
��2��N��N�ļ���Ϊ942kJ?mol-1��N-N�����ļ���Ϊ247kJ?mol-1��˵��N2�е�______����______���ȶ�����ҡ��С�����
��3��Һ̬���ɵ����NH2-��NH2-�ĵ���ʽΪ______��
��4����֪NH4HΪ���ӻ����д��������ˮD2O��Ӧ����������ȣ�______��Na3NҲΪ���ӻ������Na3N��ˮ��Ӧ�Ļ�ѧ����ʽΪ______����Ӧ����Ϊ______��
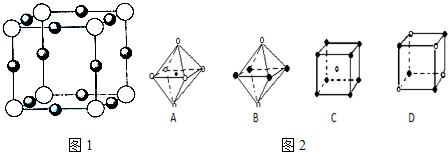
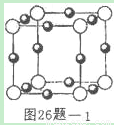
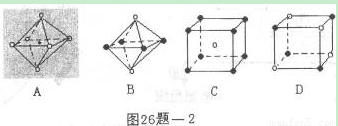
��5��X+�����е������ó���K��L��M�������Ӳ㣬����N3-�γɵľ���ṹ��ͼ1��ʾ����ͬһ��N3-������X+��______����Xԭ�ӵĻ�̬�����Ų�ʽΪ______
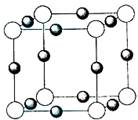
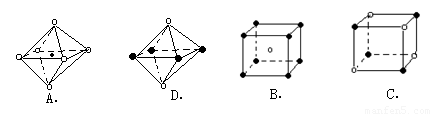
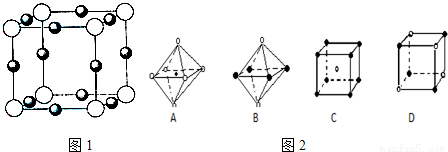
��6�������ѧ�����Ƴ�ijԪ��Z��NԪ���γɵľ���ZN����֪ZN���������NaCl���Ƶľ���ṹ��ͼ2�Ǵ�ZN����ṹͼ�зָ�����IJ��ֽṹͼ�����жϷ���ZN����ṹͼ����______��
���𰸡���������1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ���NԪ��ԭ��2p�ܼ�����3�����ӣ�Ϊ�����ȶ�״̬���������ͣ��Ƚ��ȶ���ʧȥ��һ��������Ҫ���������ࣻ
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol����1���м��ļ���Ϊ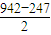 kJ/mol=347.5kJ/mol������Խ��ѧ��Խ�ȶ����Դ��жϣ�
kJ/mol=347.5kJ/mol������Խ��ѧ��Խ�ȶ����Դ��жϣ�
��3��NH2-���Կ���NH3ʧȥ1��H+��Nԭ��ԽHԭ��֮���γ�1�Թ��õ��Ӷԣ�Nԭ����2�Թ¶Ե��Ӷԣ�
��4��NH4HΪ���ӻ������笠��������⸺���ӹ��ɣ�����ˮD2O����ˮ�ⷴӦ��笠���������ˮ����������ӽ�ϣ��⸺��������ˮ����������ӽ�ϣ�
Na3NΪ���ӻ��������������N3-���ɣ���ˮ����ˮ�ⷴӦ��N3-��ˮ����������ӽ�ϣ��ݴ���д��
��5��X+�����е������ó���K��L��M�������Ӳ㣬��XԪ��ԭ�Ӻ��������Ϊ2+8+18+1=29��ΪCuԪ�أ��ɾ����ṹ��֪�������а�ɫ����ĿΪ8× =1����ɫ��Ϊ12×
=1����ɫ��Ϊ12× =3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�������ݺ�������Ų�������дCuԭ�ӵĻ�̬�����Ų�ʽ��
=3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�������ݺ�������Ų�������дCuԭ�ӵĻ�̬�����Ų�ʽ��
��6�������Ȼ��ƾ����������ӻ������ӵ���λ���ж���ṹͼ�Ƿ���ȷ�������ӻ������ӵ���λ������6��
����⣺��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ���NԪ��ԭ��2p�ܼ�����3�����ӣ�Ϊ�����ȶ�״̬���������ͣ��Ƚ��ȶ���ʧȥ��һ��������Ҫ���������࣬�ʵ�һ������N��O��C���ʴ�Ϊ��N��O��C��
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol����1���м��ļ���Ϊ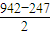 kJ/mol=347.5kJ/mol����N2�еĦм����ܴ��ڦҼ����ܣ��м����ȶ����ʴ�Ϊ���У��ң�
kJ/mol=347.5kJ/mol����N2�еĦм����ܴ��ڦҼ����ܣ��м����ȶ����ʴ�Ϊ���У��ң�
��3��NH2-���Կ���NH3ʧȥ1��H+��Nԭ��ԽHԭ��֮���γ�1�Թ��õ��Ӷԣ�Nԭ����2�Թ¶Ե��Ӷԣ�NH2-����ʽΪ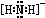 ���ʴ�Ϊ��
���ʴ�Ϊ��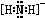 ��
��
��4��NH4HΪ���ӻ��������ˮD2O���ȷ�����ӦNH4H+D2O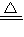 HD��+NH3��+HDO��
HD��+NH3��+HDO��
Na3NҲΪ���ӻ����Na3N��ˮ��Ӧ�Ļ�ѧ����ʽΪ Na3N+3H2O=NH3��+3NaOH������ˮ�ⷴӦ��
�ʴ�Ϊ��HD��NH3��HDO��Na3N+3H2O=NH3��+3NaOH��ˮ�ⷴӦ��
��5��X+�����е������ó���K��L��M�������Ӳ㣬��XԪ��ԭ�Ӻ��������Ϊ2+8+18+1=29��ΪCuԪ�أ��ɾ����ṹ��֪�������а�ɫ����ĿΪ8× =1����ɫ��Ϊ12×
=1����ɫ��Ϊ12× =3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�����ʹ�ͬһ��N3-������X+��6����
=3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�����ʹ�ͬһ��N3-������X+��6����
ͭΪ29��Ԫ�أ�ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104S1��
�ʴ�Ϊ��6��1s22s22p63s23p63d104S1��
��5��һ�������������ӵĸ���Ϊ12×=3�������Ӹ���Ϊ��8×=1��Ȼ��������ߵĸ���֮��Ϊ3��1��������Xn-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����C+��2������6��
��6��������NaCl�����У�ÿ��Na+��Χͬʱ����������ĵȾ����6��Cl-ͬ��ÿ��Cl-��Χͬʱ����������ĵȾ����6��Na+��ͼA����������ͼD��ѡȡ����һ�����ӣ�Ȼ����X��Y��Z�����и�õ�6���Ⱦ����������Ĵ��෴��ɵ����ӣ���������λ��Ҳ��6���ʷ����������ʴ�Ϊ��AD��
�����������ۺ��Խϴ�������ܡ���ѧ������ܹ�ϵ�����û�ѧ������д����������Ų����ɡ������ṹ�����ȣ���Ҫѧ���߱���ʵ��֪ʶ���Ѷ��еȣ�ע�⣨6����ʶ����ѧ���������ṹ��
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol����1���м��ļ���Ϊ
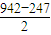 kJ/mol=347.5kJ/mol������Խ��ѧ��Խ�ȶ����Դ��жϣ�
kJ/mol=347.5kJ/mol������Խ��ѧ��Խ�ȶ����Դ��жϣ���3��NH2-���Կ���NH3ʧȥ1��H+��Nԭ��ԽHԭ��֮���γ�1�Թ��õ��Ӷԣ�Nԭ����2�Թ¶Ե��Ӷԣ�
��4��NH4HΪ���ӻ������笠��������⸺���ӹ��ɣ�����ˮD2O����ˮ�ⷴӦ��笠���������ˮ����������ӽ�ϣ��⸺��������ˮ����������ӽ�ϣ�
Na3NΪ���ӻ��������������N3-���ɣ���ˮ����ˮ�ⷴӦ��N3-��ˮ����������ӽ�ϣ��ݴ���д��
��5��X+�����е������ó���K��L��M�������Ӳ㣬��XԪ��ԭ�Ӻ��������Ϊ2+8+18+1=29��ΪCuԪ�أ��ɾ����ṹ��֪�������а�ɫ����ĿΪ8×
 =1����ɫ��Ϊ12×
=1����ɫ��Ϊ12× =3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�������ݺ�������Ų�������дCuԭ�ӵĻ�̬�����Ų�ʽ��
=3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�������ݺ�������Ų�������дCuԭ�ӵĻ�̬�����Ų�ʽ����6�������Ȼ��ƾ����������ӻ������ӵ���λ���ж���ṹͼ�Ƿ���ȷ�������ӻ������ӵ���λ������6��
����⣺��1��ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ���NԪ��ԭ��2p�ܼ�����3�����ӣ�Ϊ�����ȶ�״̬���������ͣ��Ƚ��ȶ���ʧȥ��һ��������Ҫ���������࣬�ʵ�һ������N��O��C���ʴ�Ϊ��N��O��C��
��2��N��N�к���2���м���1���Ҽ�����֪N��N����Ϊ942kJ/mol��N-N��������Ϊ247kJ/mol����1���м��ļ���Ϊ
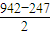 kJ/mol=347.5kJ/mol����N2�еĦм����ܴ��ڦҼ����ܣ��м����ȶ����ʴ�Ϊ���У��ң�
kJ/mol=347.5kJ/mol����N2�еĦм����ܴ��ڦҼ����ܣ��м����ȶ����ʴ�Ϊ���У��ң���3��NH2-���Կ���NH3ʧȥ1��H+��Nԭ��ԽHԭ��֮���γ�1�Թ��õ��Ӷԣ�Nԭ����2�Թ¶Ե��Ӷԣ�NH2-����ʽΪ
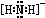 ���ʴ�Ϊ��
���ʴ�Ϊ��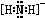 ��
����4��NH4HΪ���ӻ��������ˮD2O���ȷ�����ӦNH4H+D2O
 HD��+NH3��+HDO��
HD��+NH3��+HDO��Na3NҲΪ���ӻ����Na3N��ˮ��Ӧ�Ļ�ѧ����ʽΪ Na3N+3H2O=NH3��+3NaOH������ˮ�ⷴӦ��
�ʴ�Ϊ��HD��NH3��HDO��Na3N+3H2O=NH3��+3NaOH��ˮ�ⷴӦ��
��5��X+�����е������ó���K��L��M�������Ӳ㣬��XԪ��ԭ�Ӻ��������Ϊ2+8+18+1=29��ΪCuԪ�أ��ɾ����ṹ��֪�������а�ɫ����ĿΪ8×
 =1����ɫ��Ϊ12×
=1����ɫ��Ϊ12× =3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�����ʹ�ͬһ��N3-������X+��6����
=3���ʰ�ɫ��ΪN3-����ɫ��ΪCu+����N3-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����Cu+��2�����ʹ�ͬһ��N3-������X+��6����ͭΪ29��Ԫ�أ�ԭ�Ӻ�������Ų�ʽΪ��1s22s22p63s23p63d104S1��
�ʴ�Ϊ��6��1s22s22p63s23p63d104S1��
��5��һ�������������ӵĸ���Ϊ12×=3�������Ӹ���Ϊ��8×=1��Ȼ��������ߵĸ���֮��Ϊ3��1��������Xn-Ϊ���ģ���X��Y��Z�������ϣ���ÿ�����ϵȾ����C+��2������6��
��6��������NaCl�����У�ÿ��Na+��Χͬʱ����������ĵȾ����6��Cl-ͬ��ÿ��Cl-��Χͬʱ����������ĵȾ����6��Na+��ͼA����������ͼD��ѡȡ����һ�����ӣ�Ȼ����X��Y��Z�����и�õ�6���Ⱦ����������Ĵ��෴��ɵ����ӣ���������λ��Ҳ��6���ʷ����������ʴ�Ϊ��AD��
�����������ۺ��Խϴ�������ܡ���ѧ������ܹ�ϵ�����û�ѧ������д����������Ų����ɡ������ṹ�����ȣ���Ҫѧ���߱���ʵ��֪ʶ���Ѷ��еȣ�ע�⣨6����ʶ����ѧ���������ṹ��

��ϰ��ϵ�д�
 ��˼ά������ҵ��ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ��ټ��ִ�ѧ������ϵ�д� �����������Ż�ѧϰϵ�д�
�����������Ż�ѧϰϵ�д�
�����Ŀ