��Ŀ����
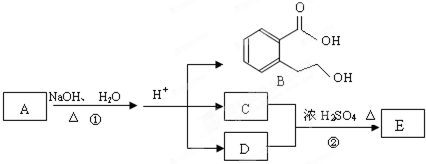
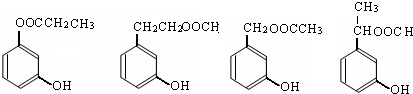
��ͼ�� A��B��C��D��E��Ϊ�л��������֪��C�ܸ�NaHCO3������Ӧ��C��D����Է���������ȣ���EΪ��֧���Ļ����

������ͼ�ش����⣺
��1����֪ E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ������E�ķ���ʽΪ__________��
��2��������B���ܷ����ķ�Ӧ��__________������ĸ��ţ���
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧ d��������Ӧ e��ˮ�ⷴӦ f�� �û���Ӧ
��3����Ӧ�ڵĻ�ѧ����ʽ��______________________________��
��4��C�����еĹ�����������__________��A�Ľṹ��ʽ��__________��
��5��ͬʱ������������������B��ͬ���칹�����Ŀ��__________����
���м��ȡ�������ṹ �����ڷǷ������� ���� FeCl3��Һ������ɫ��Ӧ��д����������һ��ͬ���칹��Ľṹ��ʽ_______________��
��1����֪ E����Է�������Ϊ102������̼��������Ԫ�ص����������ֱ�Ϊ58.8%��9.8%������Ϊ������E�ķ���ʽΪ__________��
��2��������B���ܷ����ķ�Ӧ��__________������ĸ��ţ���
a���ӳɷ�Ӧ b��ȡ����Ӧ c����ȥ��Ӧ d��������Ӧ e��ˮ�ⷴӦ f�� �û���Ӧ
��3����Ӧ�ڵĻ�ѧ����ʽ��______________________________��
��4��C�����еĹ�����������__________��A�Ľṹ��ʽ��__________��
��5��ͬʱ������������������B��ͬ���칹�����Ŀ��__________����
���м��ȡ�������ṹ �����ڷǷ������� ���� FeCl3��Һ������ɫ��Ӧ��д����������һ��ͬ���칹��Ľṹ��ʽ_______________��
��1��C5H10O2
��2��e
��3��CH3COOH + CH3CH2CH2OH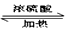 CH3COOCH2CH2CH3 + H2O
CH3COOCH2CH2CH3 + H2O
��4���Ȼ���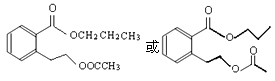
��5��4�� ��дһ�����ɣ�
��дһ�����ɣ�
��2��e
��3��CH3COOH + CH3CH2CH2OH
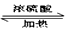 CH3COOCH2CH2CH3 + H2O
CH3COOCH2CH2CH3 + H2O ��4���Ȼ���

��5��4��
 ��дһ�����ɣ�
��дһ�����ɣ�
��ϰ��ϵ�д�
�����Ŀ

 ����1��
����1��



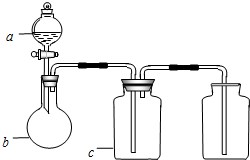 ��ͼ��a��b��c��ʾ��Ӧ�����м�����Լ���������ͼװ����ȡ���������ռ��������ǣ�������
��ͼ��a��b��c��ʾ��Ӧ�����м�����Լ���������ͼװ����ȡ���������ռ��������ǣ�������


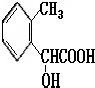
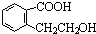
 CH2-CH2
CH2-CH2 n��CH2=CH2+H2O
n��CH2=CH2+H2O