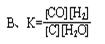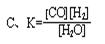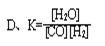��Ŀ����
N2O5��һ������������,��һ���¶��¿ɷ������з�Ӧ:2N2O5(g)  4NO2(g)+ O2(g)����H >0,T1�¶��µIJ���ʵ���������±�.����˵������ȷ����
4NO2(g)+ O2(g)����H >0,T1�¶��µIJ���ʵ���������±�.����˵������ȷ����
A.500s��N2O5�ֽ�����Ϊ2.96��10-3 mol/(L��s)
B.T1�¶��µ�ƽ�ⳣ��ΪK1 =125,1000sʱת����Ϊ50%
C.������������ʱ,T2�¶��·�Ӧ��1000sʱ���N2O5(g)Ũ��Ϊ2.98 mol/L,��T1 <T2
D.T1�¶��µ�ƽ�ⳣ��ΪK1 ,T3�¶��µ�ƽ�ⳣ��ΪK3,��K1> K3,��T1>T3
 4NO2(g)+ O2(g)����H >0,T1�¶��µIJ���ʵ���������±�.����˵������ȷ����
4NO2(g)+ O2(g)����H >0,T1�¶��µIJ���ʵ���������±�.����˵������ȷ���� | t/s | 0 | 500 | 1000 | 1500 |
| c(N2O5)mol/L | 5.00 | 3.52 | 2.50 | 2.50 |
B.T1�¶��µ�ƽ�ⳣ��ΪK1 =125,1000sʱת����Ϊ50%
C.������������ʱ,T2�¶��·�Ӧ��1000sʱ���N2O5(g)Ũ��Ϊ2.98 mol/L,��T1 <T2
D.T1�¶��µ�ƽ�ⳣ��ΪK1 ,T3�¶��µ�ƽ�ⳣ��ΪK3,��K1> K3,��T1>T3
C
2N2O5(g)  4NO2(g)+ O2(g)
4NO2(g)+ O2(g)
��ʼŨ��5.00 0 0
�仯Ũ��1.48 2.96 0.74
500sŨ��3.52
���ԣ�500s��N2O5�ֽ����� A��ȷ
A��ȷ
T1�¶���1000sʱת������Ϊ50%��
���ݷ���ʽ��2N2O5(g) 4NO2(g)+ O2(g)��
4NO2(g)+ O2(g)��
��ʼŨ�� 5.00 0 0
�仯Ũ�� 2.50 5.00 1.25
ƽ��Ũ�� 2.50 5.00 1.25
��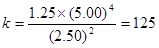 ����B��ȷ
����B��ȷ
������������ʱ,T2�¶��·�Ӧ��1000sʱ���N2O5(g)Ũ��Ϊ2.98 mol/L,����T1�¶���Ũ�ȸߣ���˵�����¶�ΪT2ʱ��ƽ�����淴Ӧ�����ƶ���������Ӧ����Ϊ���ȷ�Ӧ�����Խ���ƽ�⽫����ȷ�Ӧ���淴Ӧ�ƶ���C��
ͬ��Ҳ�ɷ���֪D��ȷ��
 4NO2(g)+ O2(g)
4NO2(g)+ O2(g)��ʼŨ��5.00 0 0
�仯Ũ��1.48 2.96 0.74
500sŨ��3.52
���ԣ�500s��N2O5�ֽ�����
 A��ȷ
A��ȷT1�¶���1000sʱת������Ϊ50%��
���ݷ���ʽ��2N2O5(g)
 4NO2(g)+ O2(g)��
4NO2(g)+ O2(g)����ʼŨ�� 5.00 0 0
�仯Ũ�� 2.50 5.00 1.25
ƽ��Ũ�� 2.50 5.00 1.25
��
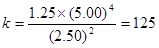 ����B��ȷ
����B��ȷ������������ʱ,T2�¶��·�Ӧ��1000sʱ���N2O5(g)Ũ��Ϊ2.98 mol/L,����T1�¶���Ũ�ȸߣ���˵�����¶�ΪT2ʱ��ƽ�����淴Ӧ�����ƶ���������Ӧ����Ϊ���ȷ�Ӧ�����Խ���ƽ�⽫����ȷ�Ӧ���淴Ӧ�ƶ���C��
ͬ��Ҳ�ɷ���֪D��ȷ��

��ϰ��ϵ�д�
�����Ŀ
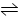 B��g��+ 2C��g�����Ҵﵽƽ�⡣�������¶�ʱ��������ܶ�������������������ȷ���ǣ� ��
B��g��+ 2C��g�����Ҵﵽƽ�⡣�������¶�ʱ��������ܶ�������������������ȷ���ǣ� ��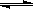 2B(g)��C(g)(����ӦΪ���ȷ�Ӧ)���ﵽƽ��ʱ��Ҫʹ�淴Ӧ���ʽ��ͣ�A��Ũ������Ӧ��ȡ�Ĵ�ʩ�� (����)
2B(g)��C(g)(����ӦΪ���ȷ�Ӧ)���ﵽƽ��ʱ��Ҫʹ�淴Ӧ���ʽ��ͣ�A��Ũ������Ӧ��ȡ�Ĵ�ʩ�� (����) 2NO(g) ��H > 0��֪�÷�Ӧ��2404��ʱ��ƽ�ⳣ��K=64��10-4����ش�
2NO(g) ��H > 0��֪�÷�Ӧ��2404��ʱ��ƽ�ⳣ��K=64��10-4����ش�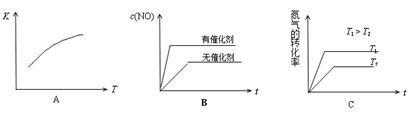
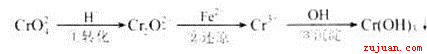
 Cr2O72������ɫ��+H2O
Cr2O72������ɫ��+H2O N2O4(g),��H<0,������ƿ����100��ķ�ˮ��,����������û�з����ı����:����ɫ����ƽ����Է�������������������ѹǿ�����ܶ�
N2O4(g),��H<0,������ƿ����100��ķ�ˮ��,����������û�з����ı����:����ɫ����ƽ����Է�������������������ѹǿ�����ܶ�  2
2 SO3(g)����H<0��ij�¶��£���2 mol SO2��1 mol O2����10 L�ܱ������У���Ӧ��ƽ���SO2��ƽ��ת����(��)����ϵ��ѹǿ(p)�Ĺ�ϵ��ͼ����ʾ��������˵����ȷ����
SO3(g)����H<0��ij�¶��£���2 mol SO2��1 mol O2����10 L�ܱ������У���Ӧ��ƽ���SO2��ƽ��ת����(��)����ϵ��ѹǿ(p)�Ĺ�ϵ��ͼ����ʾ��������˵����ȷ����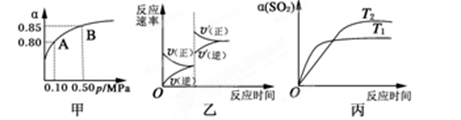
 qZ(g)����H��0��m��n��q���ں����ܱ������з�Ӧ�ﵽƽ��ʱ�������й�˵����ȷ����
qZ(g)����H��0��m��n��q���ں����ܱ������з�Ӧ�ﵽƽ��ʱ�������й�˵����ȷ���� CO(g)+H2(g)��ƽ�ⳣ��(K)��д��ʽ����ȷ���ǣ� ��
CO(g)+H2(g)��ƽ�ⳣ��(K)��д��ʽ����ȷ���ǣ� ��